Question: Consider a large furnace with opaque, diffuse, gray walls at 3000 K having an emissivity of 0.85. A small, diffuse, spectrally selective object in the
Consider a large furnace with opaque, diffuse, gray walls at 3000 K having an emissivity of 0.85. A small, diffuse, spectrally selective object in the furnace is maintained at 300 K. For the specified points on the furnace wall (A) and the object (B), indicate values for ?, ?, E, G, and J.
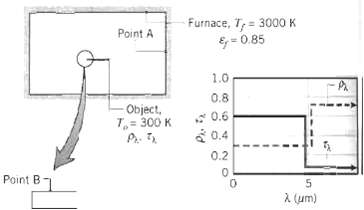
Furnace, T,= 3000 K Point A E; = 0.85 1.0 0.8 Object, 7,= 300 K * 0.6 a 0.4 0.2 Point B A (um)
Step by Step Solution
3.33 Rating (159 Votes )
There are 3 Steps involved in it
KNOWN Large furnace with diffuse opaque walls T f and a small diffuse spectrally selective object To ... View full answer

Get step-by-step solutions from verified subject matter experts
Document Format (1 attachment)
8-E-M-E-H-M-T (1142).docx
120 KBs Word File


