Which is the correct Lewis structure for nitrogen trifluoride? a) :F=N-F: F: c) :F-N-F: :F: b) :F-N-F:
Question:
Which is the correct Lewis structure for nitrogen trifluoride?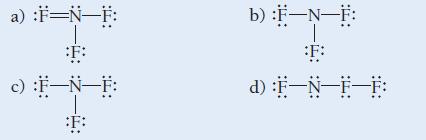
Transcribed Image Text:
a) :F=N-F: F: c) :F-N-F: :F: b) :F-N-F: F: d) :F-N-F-F:
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (2 reviews)
c...View the full answer

Answered By

JAPHETH KOGEI
Hi there. I'm here to assist you to score the highest marks on your assignments and homework. My areas of specialisation are:
Auditing, Financial Accounting, Macroeconomics, Monetary-economics, Business-administration, Advanced-accounting, Corporate Finance, Professional-accounting-ethics, Corporate governance, Financial-risk-analysis, Financial-budgeting, Corporate-social-responsibility, Statistics, Business management, logic, Critical thinking,
So, I look forward to helping you solve your academic problem.
I enjoy teaching and tutoring university and high school students. During my free time, I also read books on motivation, leadership, comedy, emotional intelligence, critical thinking, nature, human nature, innovation, persuasion, performance, negotiations, goals, power, time management, wealth, debates, sales, and finance. Additionally, I am a panellist on an FM radio program on Sunday mornings where we discuss current affairs.
I travel three times a year either to the USA, Europe and around Africa.
As a university student in the USA, I enjoyed interacting with people from different cultures and ethnic groups. Together with friends, we travelled widely in the USA and in Europe (UK, France, Denmark, Germany, Turkey, etc).
So, I look forward to tutoring you. I believe that it will be exciting to meet them.
3.00+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Draw a Lewis structure for nitrogen pentoxide (N2O5) in which each N is bonded to three O atoms.
-
Which is the correct Lewis structure for CO 3 2 - ? b) : |:0=C=0: : :: - - - := - :: :0c0: 20 -=- :: :=-
-
Which is the correct Lewis structure for magnesium bromide? a) 2 Mg+ [Br] c) Mg+2 [Br] b) Mg: Br: d) :Mg: Br:
-
help me with questions 10, 11 and 12 to answer them for practiceolin data structures Given the following linked list definitions: struct listRec! string name; listRec *link, ;: nodeType *begin; Code...
-
Harmon Household Products, Inc., manufactures a number of consumer items for general household use. One of these products, a chopping board, requires an expensive hardwood. During a recent month, the...
-
Two discrete-time systems T1 and T2 are connected in cascade to form a new system T as shown in figure. Prove or disprove the following statements. (a) If T1 and T2 are linear, than T is linear...
-
William Stem filed an action against Gary Braden, seeking to rescind a contract for the sale of an automobile and to obtain the return of the purchase price plus interest. The trial court granted...
-
Bullseye Department store is a discount retailer of general merchandise in the Southeastern United States. The company owns more than 50 stores in Florida, Georgia, South Carolina, and Tennessee that...
-
17-x, 2 for x 0 Let f(x) = Find each limit. 17+x, for x>0 (A) lim f(x) (B) lim f(x) x-0 (C) lim f(x) x-0 (D) f(0)
-
Which molecule could have an expanded octet? (a) H 2 CO 3 (b) H 3 PO 4 (c) HNO 2
-
Assign formal charges to each atom in the resonance structures of the cyanate ion (OCN ). Which resonance structure is likely to contribute most to the correct structure of (OCN )? A B C |C=N]...
-
In problem, match each graph to one the following functions. f(x) = x 2 + 2x + 2 A. B. C. D. E. F. G. H. 3 -2 -1- (-1, 0) , 2 2 -2 (-1, 1)
-
For the design described in 4.20.5, add NOPs to this instruction sequence to ensure correct execution in spite of missing support for forwarding. Problems 4.20.5 If we assume forwarding will be...
-
Discuss the distinctive features of accounting and assurance in Hong Kong under the one country two systems approach.
-
If you bought and held shares of the Priceline Group, the online provider of travel-related reservations and search services, from 2014 to 2019, you would have been one happy camper. During that...
-
Following the financial crisis of 2008, American consumers cut back on new car purchases. In response to the decline in car sales, Congress passed the program Cash for Clunkers, which allowed...
-
Find the total resistance of each group of series resistors shown in Figure 467. Data in Figure 467 . (a) ***** (b) (c) **** (d) *****
-
Montevallo Corporation leased equipment from Folio Company. The lease term is 10 years, requires payments of $25,000 at the end of each year, and contains a bargain purchase option. At the end of the...
-
What are technical skills At what level are they most important and why?
-
The pipe is subjected to a shear force of V = 8 kip. Determine the shear flow in the pipe at points A and B. 0.2 in. -6 in.
-
The stiffened beam is constructed from plates having a thickness of 0.25 in. If it is subjected to a shear of V = 8 kip, determine the shear-flow distribution in segments AB and CD. What is the...
-
The beam supports a vertical shear of V = 7 kip. Determine the resultant force in segment AB of the beam. 0.5 in. 10 in. -0.5 in. IA 0.5 in. 5 in.
-
Suppose the exchange rate is $1.59/, the euro-denominated continuously compounded interest rate is 6%, the U.S. dollar-denominated continuously compounded interest rate is 5%, and the exchange rate...
-
A random sample 20 men had their blood glucose levels (mg/100 ml) recorded after a 12 hour fast. The data are displayed in the stem-and-leaf plot below. 4 0 6 5 1 3 7 6 2 5 5 8 7 1 6 7 7 8 0 3 3 9 1...
-
What role do social control mechanisms play in regulating deviant behavior, and how do these mechanisms vary across different cultural and historical contexts ?

Study smarter with the SolutionInn App


