Consider the hypothetical eutectic phase diagram for metals A and B, which is similar to that for
Question:
Consider the hypothetical eutectic phase diagram for metals A and B, which is similar to that for the lead-tin system, Figure. Assume that
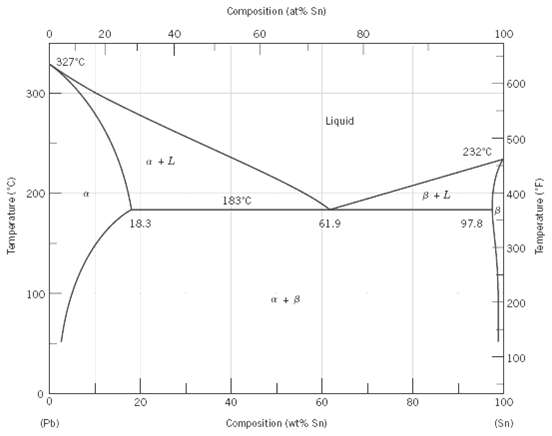
(1) ? and ? phases exist at the A and B extremities of the phase diagram, respectively;
(2) The eutectic composition is 47 wt% B-53 wt% A; and
(3) The composition of the ? phase at the eutectic temperature is 92.6 wt% B-7.4 wt% A. Determine the composition of an alloy that will yield primary ? and total ? mass fractions of 0.356 and 0.693, respectively.
Transcribed Image Text:
Composition (at% Sn) 40 20 60 80 100 327°C 600 300 Liquid 500 232°C a +L 200 400 8+L 183'C 18.3 61.9 97.8 300 100 a +8 200 100 100 20 40 60 80 Composition (wt% Sn) (Pb) (Sn) Temperature ("C) Temperature ("F)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 60% (5 reviews)
We are given a hypothetical eutectic phase diagram for which C eutectic 47 wt B C 92...View the full answer

Answered By

Muhammad Salman Alvi
Well, I am a student of Electrical Engineeing from Information Technology University of Punjab. Just getting into my final year. I have always been good at doing Mathematics, Physics, hardware and technical subjects. Teaching profession requires a alot of responsibilities and challenges.
My teaching experience started as an home tutor a year ago. When I started teaching mathematics and physic subjects to an O Level student. He was about 14 years old. His name was Ibrahim and I used to teach him for about 2 hours daily. Teaching him required a lot of patience but I had to be polite with him. I used to give him a 5 min break after 1 hour session. He was quite weak in basic maths and calculation. He used to do quite a lot of mistakes in his homework which I gave him weekly. So I decided to teach him basics from scratch. He used to say that he got the concept even if he didn't. So I had to ask him again and again. I worked on his basics for a month and after that I started taking a weekly test sesions. After few months he started to improve gradually. Now after teaching him for about a year I can proudly say that he has improved alot. The most important thing was he managed to communicate all the difficullties he was facing. He was quite capable and patient. I had a sincere desire to help him reach to its full potential. So I managed to do that. We had a very good honest relationship of a student and a teacher. I loved teaching him as a tutor. Now having an experience of one year teaching I can read students quite well. I look forward to work as an online tutor who could help students in solving their all sort of difficulties, problems and queries.
4.90+
29+ Reviews
43+ Question Solved
Related Book For 

Fundamentals of Materials Science and Engineering An Integrated Approach
ISBN: 978-1118061602
4th Edition
Authors: David G. Rethwisch
Question Posted:
Students also viewed these Materials Science Engineering questions
-
Consider a hypothetical eutectic phase diagram for metals A and B that is similar to that for the lead-tin system (Figure 9.8). Assume that: (1) and phases exist at the A and B extremes of the...
-
Construct the hypothetical phase diagram for metals A and B between temperatures of 600C and 1000C given the following information: The melting temperature of metal A is 940C. The solubility of B...
-
Construct the hypothetical phase diagram for metals A and B between temperatures of 600C and 1000C given the following information: The melting temperature of metal A is 940C. The solubility of B...
-
According to the Statute of Frauds, in order to be legally enforceable, a contract must be in writing, name the contracting parties, identify the subject matter of the contract, and Be for a legal...
-
Locate the liabilities section of the balance sheet of both firms, Walmart and Target. a.) For the most current year, what percent of the liabilities are short-term liabilities? Which firm has the...
-
An incomplete cost of goods manufactured schedule is presented below. Instructions Complete the cost of goods manufactured schedule for Hobbit Company. BIT COMPANY Cost of Goods Manufactured Schedule...
-
An observatory has a telescope that requires an initial investment of $\$ 25,000$. A benefactor will provide a $\$ 7,500$ gift every 5 years as long as the university makes the telescope available to...
-
One ton of liquid water at 80°C is brought into a well-insulated and well-sealed 4-m x 5-m x 7-m room initially at 22°C and 100 kPa. Assuming constant specific heats for both air and water at...
-
In the context of global interconnectedness, how should we approach the ethical responsibilities of individuals, corporations, and governments towards marginalized communities and vulnerable...
-
On April 1, 2015, Benton Corporation purchased 80% of the outstanding stock of Crandel Company for $425,000. A condensed balance sheet of Crandel Company at the purchase date is shown below. All book...
-
The microstructure of a lead-tin alloy at 180oC (355oF) consists of primary and eutectic structures. If the mass fractions of these two micro constituents are 0.57 and 0.43, respectively, determine...
-
For an 85 wt% Pb-15 wt% Mg alloy, make schematic sketches of the microstructure that would be observed for conditions of very slow cooling at the following temperatures: 600C (1110F), 500C (930F),...
-
Solid-state mass storage like a flash drive is plugged into a computer through a(n) ____________ port.
-
As noted earlier, a planned court system that is similar to the Ohio commercial docket is the proposed science court. The science court would act as a forum for cases involving scientific and...
-
Define and distinguish between future value and present value.
-
What are the influences on the supply of U.S. dollars in the foreign exchange market?
-
Colombia is the worlds biggest producer of roses. The global demand for roses increases and at the same time Columbias central bank increases the interest rate. In the foreign exchange market for...
-
How is the supply of money determined and how does it depend on the Feds monetary policy strategy?
-
What is the equity multiplier?
-
Write electron configurations for the following ions, and determine which have noble-gas configurations: (a) Cd2+ (b) p3- (c) Zr4+ (d) Ru3+ (e) As3- (f) Ag+
-
A calorimeter contained 75.0 g of water at 16.95C. A 93.3-g sample of iron at 65.58C was placed in it, giving a final temperature of 19.68C for the system. Calculate the heat capacity of the...
-
An undeformed specimen of some alloy has an average grain diameter of 0.050 mm. You are asked to reduce its average grain diameter to 0.020 mm. Is this possible? If so, explain the procedures you...
-
A non-cold-worked brass specimen of average grain size 0.01 mm has a yield strength of 150 MPa (21,750 psi). Estimate the yield strength of this alloy after it has been heated to 500C for 1000 s, if...
-
The following yield strength, grain diameter, and heat treatment time (for grain growth) data were gathered for an iron specimen that was heat treated at 800(C. Using these data compute the yield...
-
You are the merchandise manager for Best Buy electronics and have been asked to expand the assortment of music products. Industry trends suggest more people are downloading their music online. How...
-
What impact has incurring major external debt by LDCs had on global and country income distribution? What impact have attempts to reduce the debt crisis had on income distribution?
-
What are the signs of potential aspiration in: (i) the medical history, (ii) parent case history, and (iii) clinical observation? How does this differ in a premature infant compared with a term...

Study smarter with the SolutionInn App


