Convert the following Fischer projections into tetrahedral representations, and assign R or S stereochemistry toeach: H (b)
Question:
Convert the following Fischer projections into tetrahedral representations, and assign R or S stereochemistry toeach:
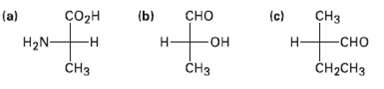
Transcribed Image Text:
сHз (b) (a) H2N- сорн -н сно (c) -Он —он -сно Н CH-CHз CHз CHз т
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 75% (12 reviews)
Horizontal bonds of Fischer projections point o...View the full answer

Answered By

Charles Okinda
students should give all the instructions concerning the challenge that they face. they will get an immediate response because I am always online.
4.90+
753+ Reviews
1480+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Convert the following Fischer projections to perspective formulas. (a) (b) (c) (d) H,N- H- CH;
-
Name and assign R or S stereochemistry to the product(s) you would obtain by reaction of the following substance with ethyl magnesium bromide. Is the product chiral? Is it optically active? Explain.
-
Assign R or S stereochemistry to the chirality centers in the following Newmanprojections: CI (a) (b) C "CH . "
-
Krell Industries has a share price of $22 46 today. If Krell is expected to pay a dividend of S0.83 this year, and its stock price is expected to grow to $24.11 at the end of the year, what is...
-
If you were a team leader at a summer camp for children with special needs, should you include this in your employment history if you are applying for work that is unrelated? Explain your answer.
-
Will Googles pay strategy work forever? Consider the evolution of Microsofts pay strategy as its growth slackened. Should Google prepare for a similar future? If so, when and what sort of actions...
-
A mountain chart is a creative way to display the distribution of a quantitative variable over different categories. The overall distribution is shown as a smoothed histogram, and the area underneath...
-
Bailey Corporation issued 10,000 shares of no-par common stock for $25 per share. Bailey also issued 3,000 shares of $40 par, 6 percent noncumulative preferred stock at $42 per share. Required a....
-
SOUTH DAKOTA CORPORATION SOUTH DAKOTA CORPORATION uses a job - order costing system with normal costing. At the start of the period, the company had the following balances: Raw Materials inventory $...
-
The University of the Bahamas has two operating departments: School of Business and School of Education. Each of the operating departments uses the services of the company's two support departments:...
-
Classify each of the following monosaccharide?s: la) 0 (c) (b) (d) CH- . H- C=0 C=0 -- -- -- -- -- -- -- -- -- H2OH -- CH- Ribulose CH Threose CH- Tagatose 2-Deoxyribose
-
Which of the following Fischer projections of glyceraldehydes represent the sameenantiomer? CH- - 3 - -CH- - - CH2 A
-
Use the following information to prepare a classified balance sheet for Alpha Co. at the end of 2018. Accounts receivable............................$26,500 Accounts...
-
Read this case study and then respond with how you would handle this situation: Randy is a person who frequently offends others due to his honest and open expression. He oftentimes says things...
-
Electrical disturbances travel at the speed of light, and radio waves differ from visible light only in the frequency at which they oscillate. The speed of light is designated by "c", and is part of...
-
In the laboratory a student finds that it takes 83.9 Joules to increase the temperature of 13.5 grams of solid iron from 24.4 to 39.2 degrees Celsius. The specific heat of iron he has measured is...
-
A chunk of nickel weighing 19.0 grams and originally at 98.12 C is dropped into an insulated cup containing 83.6 grams of water at 21.62 C. Assuming that all of the heat is transferred to the water,...
-
What is the frequency of light with a wavelength of 756nm?
-
The Swanlake Tax Services business was established on 1 March of the current year to help clients with their tax planning. During January, the business entered into the following transactions:...
-
Heineken N.V., a global brewer based in the Netherlands, reports the following balance sheet accounts for the year ended December 31, 2016 (euros in millions). Prepare the balance sheet for this...
-
Find the pH of a 0.100 M NaCHO 2 solution. The salt completely dissociates into Na + (aq) and CHO 2 (aq), and the Na + ion has no acid or base properties.
-
How does the resonance model for benzene explain the fact that there are only three isomers of dibromobenzene?
-
Which compound is more reactive toward electrophilic substitution (for example, nitration)? OCH or b. CH,CH3 a. ETor
-
The explosive TNT (2,4,6-trinitrotoluene) can be made by nitrating toluene with a mixture of nitric and sulfuric acids, but the reaction conditions must gradually be made more severe as the nitration...
-
We manufacture breakfast cereal at our factory in Toad Suck, AR. Our current selling price is $2.40 per box, F.O.B. our shipping dock (our customers pay shipping expenses). Our gross profit margin is...
-
exploration of the history of street art and its controversies of ppt/quicktime video.
-
Solve the following problems. Reduce answers to their lowest terms. Box your answers. 1 5 1.+ -= 4 12 2 2.- + 3 9 = 11 Convert improper fractions to mixed numbers. 2 11 3. + == 5 12 4 4 4. 12 7 5.35...

Study smarter with the SolutionInn App


