Question: Determine algorithms for carrying out the following flash calculations, assuming that expressions for K-values and enthalpies areavailable. Find Given he. P hs. T T.P hs,
Determine algorithms for carrying out the following flash calculations, assuming that expressions for K-values and enthalpies areavailable.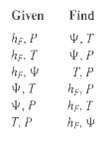
Find Given he. P hs. T T.P hs, P hs. T .T V. P T. P
Step by Step Solution
3.46 Rating (166 Votes )
There are 3 Steps involved in it
The equations to be solved for each algorithm are those for the standard adi... View full answer

Get step-by-step solutions from verified subject matter experts
Document Format (1 attachment)
37-E-C-E-S-P (133).docx
120 KBs Word File


