Diazepam, marketed as an antianxiety medication under the name Valium, has three rings, eight double bonds, and
Question:
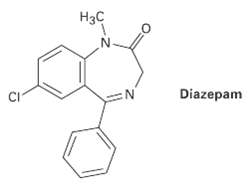
Diazepam, marketed as an antianxiety medication under the name Valium, has three rings, eight double bonds, and the formula C16H??C1N2O. How many types of hydrogen does diazepam have? (Calculate the answer; don?t count hydrogens in the structure)
Transcribed Image Text:
Нзс Diazepam
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (6 reviews)
A C16 hydrocarbon with 11 degrees of unsaturation three ...View the full answer

Answered By

Larlyu mosoti
I am a professional writer willing to do several tasks free from plagiarism, grammatical errors and submit them in time. I love to do academic writing and client satisfaction is my priority. I am skilled in writing formats APA, MLA, Chicago, and Harvard I am a statistics scientist and I can help out in analyzing your data. I am okay with SPSS, EVIEWS, MS excel, and STATA data analyzing tools.
Statistical techniques: I can do linear regression, time series analysis, logistic regression, and some basic statistical calculations like probability distributions. . I'm ready for your working projects!
Services I would offer:
• Academic writing.
• Article writing.
• Data entry.
• PDF conversion.
• Word conversion
• Proofreading.
• Rewriting.
• Data analyzing.
The best reason to hire me:
- Professional and Unique work in writing.
- 100% satisfaction Guaranteed
- within required time Express delivery
- My work is plagiarism Free
- Great communication
My passion is to write vibrantly with dedication. I am loyal and confident to give my support to every client. Because Client satisfaction is much more important to me than the payment amount. A healthy client-contractor relationship benefits in the longer term. Simply inbox me if you want clean work.
5.00+
3+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
How many types of atoms can you expect to find in a pure sample of any element?
-
Loratadine, marketed as an antiallergy medication under the name Claritin, has four rings, eight double bonds, and the formula C 22 H ? C1N 2 O 2 . How many types of hydrogen does loratadine have?...
-
How many types of hydrogen does each of the following compounds have? (a) C8H? O2, has two rings and one double bond (b) C7H? N, has two double bonds (c) C911? NO, have one ring and three double bonds
-
Four wooden beams, each of length 2a, are nailed together at their midpoints to form the support system shown. Assuming that only vertical forces are exerted at the connections, determine the...
-
1. To what extent do you think Dan Dienst was facing a crisis leadership situation when he was appointed to the CEO of the Martha Stewart Company? 2. So what leadership steps should Dienst take to...
-
The frame supports the triangular distributed load shown. Determine the normal and shear stresses at point D that act perpendicular and parallel, respectively, to the grains. The grains at this point...
-
Based on your experiences in college, draw and label a conceptual model that shows the factors that lead to your satisfaction (or dissatisfaction) with a course?
-
New employees at Sun Microsystems begin their orientation sessions after being hired with a computer game. It is part of an attempt to integrate new people, improve the image of the company, get...
-
Please provide a short paragraph response for each of the questions below. Do you think it's right or justified to have an unregulated Marketing Research industry? How do you see the role of...
-
Bruce also requires a calculation of taxable income for the most recent year ended December 31, 2019. The calculation should be prepared in Excel. He has also requested that you prepare the year-end...
-
Calculate the degree of unsaturation in the following formulas: (a) C 6 H 5 N (b) C 6 H 5 NO 2 (c) C 8 H 9 C 13 (d) C 9 H 16 Br 2 (e) C 10 H 12 N 2 O 3 (f) C 20 H 32 C1N
-
Give IUPAC names for the following compounds: CH CH3CH2CH=CCH2CH3 (b) (a) CH CHCH-H CHCH2CH2CHCHCHCH2CH3 CH CH (d) (c) CHCHCHCHC CH
-
Doggy World operates a chain of pet stores in the Midwest. The manager of each store reports to the regional manager, who, in turn, reports to the headquarters in Milwaukee, Wisconsin. The actual...
-
W has knee surgery at the hospital where she works and the hospital discounts her bill by $2,000, as it does for all employees. This is 20% of the amount of the hospital's usual charge for surgery....
-
To be useful in financial statements analysis, a ratio must meet what two conditions? Elaborate Robustness and fruitfulness?
-
A patient is unable to pay their copayment in full. Collect what they can pay and discuss payment options for the remainder
-
What is the JAPAN AGREEMENT ON COMPUTER PRODUCTS AND SERVICES? Explain briefly
-
Water Source Inc. manufactures badminton rackets. The company estimates the following costs for the next year: Indirect factory wages $171,000 Supervisor salaries 42,000 Direct materials 213,000...
-
Draw Bode plots for a sixth-order Pade approximation to a pure delay of 1 second.
-
For the next several days, take notes on your listening performance during at least a half-dozen situations in class, during social activities, and at work, if applicable. Referring to the traits of...
-
Water is a good solvent for many substances. What is the molecular basis for this property, and why is it significant?
-
Although compounds are indexed by their IUPAC substitutive names, sometimes chemists give whimsical names to compounds that they discover. Assist these two chemists by providing substitutive names...
-
Although compounds are indexed by their IUPAC substitutive names, sometimes chemists give whimsical names to compounds that they discover. Assist these two chemists by providing substitutive names...
-
Within each set, which two structures represent the same compound? A B
-
Explore the concept of principal quantum numbers in quantum mechanics and atomic structure. How do principal quantum numbers, denoted by the symbol "n," define the energy levels and electron...
-
1. How many electrons make up a charge of 1.0 MC? 2. What is the total charge on 1.0 kg of electrons?
-
What frequency of sound would have a wavelength the same size as a 0.81 m -wide window? (The speed of sound is 344m/s at 20 o C .)

Study smarter with the SolutionInn App


