Loratadine, marketed as an antiallergy medication under the name Claritin, has four rings, eight double bonds, and
Question:
Loratadine, marketed as an antiallergy medication under the name Claritin, has four rings, eight double bonds, and the formula C22H? C1N2O2. How many types of hydrogen does loratadine have? (Calculate your answer; don?t count hydrogens in the structure.)
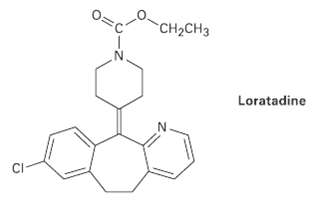
Transcribed Image Text:
CH2CH3 Loratadine
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 60% (15 reviews)
Solve this problem in the same way as we solved problems A C22 hydr...View the full answer

Answered By

Sarah Khan
My core expertise are:
-_ Finance
-_ Business
-_ Management
-_ Marketing Management
-_ Financial Management
-_ Corporate Finance
-_ HRM etc...
I have 7+ years of experience as an online tutor. I have hands-on experience in handling:
-_ Academic Papers
-_ Research Paper
-_ Dissertation Paper
-_ Case study analysis
-_ Research Proposals
-_ Business Plan
-_ Complexed financial calculations in excel
-_ Home Work Assistance
-_ PPT
-_ Thesis Paper
-_ Capstone Papers
-_ Essay Writing etc...
5.00+
91+ Reviews
92+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
How many types of atoms can you expect to find in a pure sample of any element?
-
Diazepam, marketed as an antianxiety medication under the name Valium, has three rings, eight double bonds, and the formula C 16 H ?? C1N 2 O. How many types of hydrogen does diazepam have?...
-
How many types of hydrogen does each of the following compounds have? (a) C8H? O2, has two rings and one double bond (b) C7H? N, has two double bonds (c) C911? NO, have one ring and three double bonds
-
Select appropriate letter to each of its related definition in the list given below. Definition A. Economic resources to be used or turned into cash within one year. B. Reports assets, liabilities,...
-
Several people have commented that this chapter deals with the "nuts and bolts" of leadership. What makes them say that?
-
Consider a 16-bit processor in which the following appears in main memory, starting at location 200: The first part of the first word indicates that this instruction loads a value into an...
-
What conditions must be met for stud mounting an accelerometer?
-
The ledger of Clayton Company includes the following unadjusted balances: Prepaid Insurance $3,000, Service Revenue $58,000, and Salaries and Wages Expense $25,000. Adjusting entries are required for...
-
Molin Corporation is a manufacturer that uses job-order costing. The company closes out any overapplied or underapplied overhead to Cost of Goods Sold at the end of the year. The company has supplied...
-
The Chestnut Street Company plans to issue a bond semiannually on March 31st and September 30th. The Controller has asked you to calculate information about the bond assuming two different market...
-
Calculate the degree of unsaturation in the following formulas, and draw five possible structures for each: (a) C10H16 (b) C8H8O (c) C7H10C12 (d) C10H16O2 (e) C5H9NO2 (f) C8H10C1NO
-
Name the following alkenes: ) CH2H2CH {c) CH-CH (a) H (b) -CH CHCH2CH3 CH H%3H2H H3C (9) H%3D%3H3 (e) CH (d) C=C 23DCHCHCH CH CH CH CHCH2CH2
-
For each of the two (separate) experiments that follow, perform the Wilcoxon test. If an experiment yields statistically significant results, also compute r c . (b) Experiment 4 X1 (a) Experiment 3...
-
The so-called newsvendor model compares the net salvage cost to the (opportunity) cost of stocking out (this opportunity cost is typically the unit contribution margin) and tells us whether to order...
-
Earlier this year, University of California - Los Angeles (UCLA) & the University of Southern California (USC) announced that they would be leaving the Pacific 12 Conference(PAC 12) for the Big Ten...
-
Who has the power to make an offer? If a statement or promise is made, does it constitute a good offer, using the elements of the QQC test (quality, quantity, communication)?
-
Janz v Whitehorse Wholesale Auto Centre Ltd., 2016 YKSC 27 (CanLII) Janz entered into two lease/purchase agreements with Whitehorse for two trucks. She did not make several of the payments required...
-
Michael Irvin, former Dallas Cowboy & current commentator for NFL Network, is suing Marriott Hotels and one employee for defamation. Irvin's lawsuit alleges he has been wrongfully accused of sexual...
-
Look carefully at the wording of the debit entry for items 11 and 12. What is being debited, the amount due, or the people who owe the business these amounts?
-
How much more interest will be earned if $5000 is invested for 6 years at 7% compounded continuously, instead of at 7% compounded quarterly?
-
Spilling room-temperature water over your skin on a hot day cools you down. Spilling room-temperature vegetable oil over your skin on a hot day does not. Explain the difference.
-
Why isn't the alkene starting material in Eq. 4.43 part of the equilibrium mixture?
-
Isopropyl alcohol is produced commercially by the hydration of propene. Show the mechanistic steps of this process. If you do not know the structure of isopropyl alcohol, try to deduce it by analogy...
-
Give the structures and the IUPAC substitutive names of the isomeric alkenes with the molecular formula CuH,, containing four carbons in their principal chain.
-
Jamie Lee is attracted to the low monthly payment advertised for a vehicle lease. She may well be able to afford a more expensive car than she originally thought. Jamie Lee really needs to think this...
-
Explain virtual hosting and how to use either name-based or IP-based virtual hosting.?
-
Discuss the role of literary allusion in satire as a means of subverting conventions and critiquing societal norms.

Study smarter with the SolutionInn App


