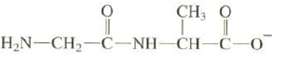
Di-peptides result from the reaction of two amino acids to form an amide. Explain which nitrogen of
Question:
Di-peptides result from the reaction of two amino acids to form an amide. Explain which nitrogen of the following di-peptide is the stronger base:
Transcribed Image Text:
CH,O LI H_N–CH, C-NH-CH-C-0
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 64% (14 reviews)
The N that is bonded to the C O group is not very basic beca...View the full answer

Answered By

Chiranjib Thakur
I have no tutoring experience yet, but I can share my skills and knowledge gained from my education and work experiences. I have been a CPA since 2012 with 6 years of work experience in internal auditing and 4 years of work experience in accounting at the supervisory level.
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Two substitution products result from the reaction of 3-chloro-3-methyl-1-butene with sodium acetate (CH3COO-Na+) in acetic acid under SN1 conditions. Identify the products
-
Two products are obtained from the reaction of (Z)-2-pentene with water and a trace of H2SO4. The mass spectra of these products are shown in Figure 13.10. Identify the compounds responsible for the...
-
Two stereoisomers are obtained from the reaction of HBr with (S)-4-bromo-1-pentene. One of the stereoisomers is optically active, and the other is not. Give the structures of the stereoisomers,...
-
Recent trends in recruiting rely on social capital by ______. Multiple choice question. making use of structural holes between groups in a social network where there are few relationships bridging...
-
What is the meaning of the term interaction between variances?
-
What is endogenous growth theory and what puzzles associated with the Solow growth model does it attempt to resolve?
-
Air flows past two equal sized spheres (one rough, one smooth) that are attached to the arm of a balance as is indicated in Fig. P9.87. With \(U=0\) the beam is balanced. What is the minimum air...
-
Refer to the Buena School District bus data. a. Develop a 95% confidence interval for the mean bus maintenance. b. Develop a 95% confidence interval for the mean bus miles.
-
Should southwest airlines - use all debt, all stock, or a 50-50 combination of debt and stock to finance future market-development strategy? Provide a summary recommendation/analysis overview of the...
-
What is output of the following program? #include using namespace std; int x = 19; int main () { int x = 21; { int x = 41; cout <
-
Amino acids contain both a basic functional group, the amine, and an acidic functional group, the carboxylic acid. Thus, they can undergo an internal acid-base re-action as shown in the following...
-
Explain which nitrogen in the ring of the amino acid histidine is the stronger base: N- N-H CH + HN CHCO, Histidine
-
Let M(x) be a companys marginal profit at production level x. Give an economic interpretation of the number 44 48 M(x)dx.
-
Describe why healthcare decisions can change as circumstances change.
-
Describe what is meant by the term political malpractice.
-
Describe the consultative role of the ethics committee when addressing ethical dilemmas.
-
Discuss the importance of duty to warn for behavioral health professionals.
-
Describe how spirituality and religion help to shape ones healthcare choices.
-
What is the effect on the current through a circuit of steady resistance when the voltage is doubled? What is the effect on the current if both voltage and resistance are doubled?
-
U.S. households have become smaller over the years. The following table from the 2010 GSS contains information on the number of people currently aged 18 years or older living in a respondent's...
-
Sketch the graph of the equation. y = 2
-
The pKa of p-cyclopropylbenzoic acid is 4.45. Is cyclopropylbenzene likely to be more reactive or less reactive than benzene toward electrophilic bromination? Explain.
-
Rank the following compounds in order of increasing acidity. Dont look at a table Of PKa data to help with your answer. (a) Benzoic acid, p-methyl benzoic acid, p-chlorohenzoic acid (b)...
-
How would you prepare the following carboxylic acids? (a) (CH 3 ) 3 CCO 2 H from (CH 3 ) 3 CCl (b) CH 3 CH 2 CH 2 CO2H from CH 3 CH 2 CH 2 Br
-
Assume that a robot systems constant failure and repair rates are 0.0004 failures per hour and 0.002 repairs per hour, respectively. Calculate the robot system steady-state unavailability and...
-
March 6, 2018 David Beckham's Latest Pitch: Men's Cosmetics. By Robert Williams Men of the world, David Beckham has a message for you: Moisturize. "It's the biggest thing," he says in a London...
-
3. On January 1, an investment fund was opened with an initial balance of 5000. Just after the balance grew to 5200 on July 1, an additional 2600 was deposited. The annual effective yield rate for...

Study smarter with the SolutionInn App


