Draw the product from reaction of each of the following substances with (i) Br2, FeBr3 and (ii)
Question:
Draw the product from reaction of each of the following substances with (i) Br2, FeBr3 and (ii) CH3COCl,AlCl3.
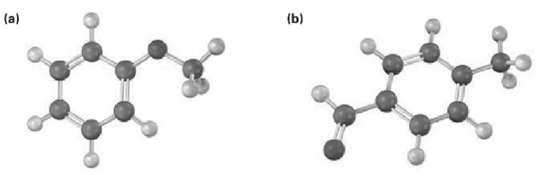
Transcribed Image Text:
(b) (a)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 57% (14 reviews)
a The methoxyl group is an orthopara director OCH3 OCH3 Br FeBr3 Br pBromome...View the full answer

Answered By

Utsab mitra
I have the expertise to deliver these subjects to college and higher-level students. The services would involve only solving assignments, homework help, and others.
I have experience in delivering these subjects for the last 6 years on a freelancing basis in different companies around the globe. I am CMA certified and CGMA UK. I have professional experience of 18 years in the industry involved in the manufacturing company and IT implementation experience of over 12 years.
I have delivered this help to students effortlessly, which is essential to give the students a good grade in their studies.
3.50+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Give the major product obtained from reaction of each of the following with excess HCl: a. CH3CH2C==CH b. CH3CH2C===CCH2CH3 c. CH3CH2C==CCH2CH2CH3
-
Predict the product from reaction of the following substance (reddish brown = Br) with: (a) PBr3 (b) Aqueous H2SO4 (c) SOCl2 (d) PCC (e) Br2,FeBr3
-
Predict the product from reaction of the following substance with: (a) NaBH4 then H3O+ (b) LiAlH4 then H3O+ (c) CH3CH2 MgBr; then H3O+
-
What is the probability that the number X of successes in the personnel managers sample of 100 employees in question 10 will be 48 or more?
-
In reporting the transactions of a self-employed taxpayer, when can a Schedule C-EZ be used instead of the regular Schedule C of Form 1040?
-
Nickel has two major and three minor isotopes. For the purpose of this problem, suppose that the only isotopes are 58 Ni and 60 Ni. The atomic mass of 58 Ni is 57.935 3 Da and the mass of 60Ni is...
-
P(170 < x < 195) The random variable x is normally distributed with mean = 174 and standard deviation = 20. Find the indicated probability.
-
Each day Seacoast Food Services makes deliveries to four restaurants it supplies in the metro Atlanta area. The service uses one truck that starts at its warehouse, makes a delivery to each...
-
Would others view you as an ethical leader? Please explain what traits you possess or behaviors you demonstrate (or the lack of) to support your reasoning of being an ethical leader. Please provide...
-
On January 1, 2020, Paloma Corporation exchanged $1,710,000 cash for 90 percent of the outstanding voting stock of San Marco Company. The consideration transferred by Paloma provided a reasonable...
-
In planning synthesis, it?s as important to know what not do as to know what to do. As written, the following reaction schemes have flaws in them. What is wrong with each? (a) CN CN 1. CH3CH,COCI,...
-
The following molecular model of a dimethyl-substituted biphenyl represents the lowest-energy conformation of the molecule. Why are the two benzene rings tilted at a 63 o angle to each other rather...
-
A \(0.20-\mathrm{kg}\) softball is traveling at a velocity of \(20 \mathrm{~m} / \mathrm{s}\) to the east relative to Earth. It collides head-on with a 0. 40 \(\mathrm{kg}\) rubber ball traveling at...
-
An employee who oversees the counting and depositing cash holdings at end of the day urgently needs some extra cash to purchase new tires for her car. What is the part of fraud triangle does this...
-
What does Hofstede's Uncertainty Avoidance Dimension refer?
-
You, CPA, are the auditor of Sticks and Stones Ltd. (SSL), whose year end is December 31, Year 1. At year end SSL has accrued a provision of $100,000 related to an ongoing lawsuit and has included a...
-
What does it mean that political campaigns are candidate-centered?
-
Financial information is presented here for two companies. Fill in the missing amounts. (Do not express any of the numbers in parenthesis.) Young Ltd. Sales: $90,000 Sales Returns: ? Net Sales:...
-
Compared with broad-market-cap weighting, the international equity strategy suggested by McMahon is most likely to: A. concentrate risk exposure. B. be based on the efficient market hypothesis. C....
-
If a and b are positive numbers, find the maximum value of f ( x ) = x a (9 x ) b on the interval 0 x 9.
-
A tank containing carbon dioxide at 400 K and 50 bar is vented until the temperature in the tank falls to 300 K. Assuming there is no heat transfer between the gas and the tank, find the pressure in...
-
Explain why only one of the two chlorines of 1, 2-dichloro-2-methylpropane is replaced by a hydroxy group when the compound is heated in water (see the preceding hydrolysis reaction.
-
On the basis of the bond cleavage shown for this reaction in Figure 10.1, predict the stereo chemistry of the product.Explain. OCCH, CH,CH, ." -
-
Show the products of thesereactions: CI CH3CO, NaOH a) DMSO . Br CH,CO, b) DMF CH
-
Oak Creek Furniture Factory (OCFF), a custom furniture manufacturer, uses job order costing to track the cost of each customer order. On March 1, OCFF had two jobs in process with the following...
-
Image transcription text 9:10 X Test_Belgeleme_Vak... Q E .. . Yapisal Test ve Validasyon Takimi Test Belgeleme Muhendisligi Aday Vaka Calismasi Ad: Soyad: 1) Asagidaki sekilde, kok noktasindan...
-
Individual Key Learning Report Individual Key Learning Report A+ Individual Report on Key Learnings Students are required to prepare an individual report at the end of the semester to present the...

Study smarter with the SolutionInn App


