Each of the following syntheses requires more than one step. How would you carry themout? (a) CH3CH2CH2C=CH
Question:
Each of the following syntheses requires more than one step. How would you carry themout?
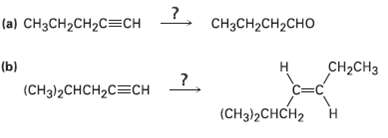
Transcribed Image Text:
(a) CH3CH2CH2C=CH CH3CH2CH2CHO (b) (CHз)2CHCH2CСH CH-CHз н C=C (CHд)2CHCH2 ?. Н
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 57% (14 reviews)
a b H Lindlar catalyst CH3CHCH...View the full answer

Answered By

Deepak Pal
Hi there! Are you looking for a committed, reliable, and enthusiastic tutor? Well, teaching and learning are more of a second nature to me, having been raised by parents who are both teachers. I have done plenty of studying and lots of learning on many exciting and challenging topics. All these experiences have influenced my decision to take on the teaching role in various capacities. As a tutor, I am looking forward to getting to understand your needs and helping you achieve your academic goals. I'm highly flexible and contactable. I am available to work on short notice since I only prefer to work with very small and select groups of students. Areas of interest: Business, accounting, Project management, sociology, technology, computers, English, linguistics, media, philosophy, political science, statistics, data science, Excel, psychology, art, history, health education, gender studies, cultural studies, ethics, religion. I am also decent with math(s) & Programming. If you have a project you think I can take on, please feel welcome to invite me, and I'm going to check it out!
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
How would you carry out the following conversions? More than one step is needed in eachcase. "
-
Syntheses of each of the following compounds have been reported in the chemical literature. Using the indicated starting material and any necessary organic or inorganic reagents, describe short...
-
Syntheses of each of the following compounds have been reported in the chemical literature. Using the indicated starting material and any necessary organic or inorganic reagents, describe short...
-
Spencer Company sells lamps and other lighting fixtures. The purchasing department manager prepared the following inventory purchases budget. Spencers policy is to maintain an ending inventory...
-
In the previous problem, suppose that vendor A provides a new price-discount schedule for component 3. This one is an "incremental" discount, as opposed to an "all-units" discount, as follows: Unit...
-
The data show the scores of the winning teams in the Rose Bowl. Construct a frequency distribution for the data using a class width of 7. 38 20 45 24 21 26 38 49 32 41 41 34 37 34 17 38 21 20 41 38...
-
A faculty member wishes to assess the relationship between students' scores on the Scholastic Aptitude test (SAT) and their performance in college. a. What is a possible research hypothesis in this...
-
Applying the net present value approach with and without tax considerations Luther Currie, the president of Luther's Moving Services, Inc., is planning to spend $625,000 for new trucks. He expects...
-
How do advanced risk aggregation techniques, such as nested simulation or extreme value theory, address the challenges posed by non-linear interactions and tail dependencies in multi-asset portfolios?
-
Suppose David is a busy CEO and has high levels of consumption (C) but relatively little leisure time (L). Mike, on the other hand, has a part-time job and therefore lots of leisure (L) with...
-
Propose structures for hydrocarbons that give the following products on oxidative cleavage by KMnO4 orO3: (b) .Co2 (a) O2 + CH(CH2)5C02H CC2H + e) O2CICH2)8CO2H (d) CH + 2H2cO2H + co2 (e) CO2...
-
How would you carry out the following transformation? More than one step isneeded. CH3CH2CH2CH2C=CH T ? CCH2CH2CH2 H
-
On December 1, 2018, Joy Vasek Importers Inc. borrowed US$1,000,000 from its bank. The loan is payable in full on July 1, 2019. Interest is payable each month end at 5% per annum. Pertinent exchange...
-
Face amount of bond: $ 1 , 0 0 0 Terms of redemption: $ 1 , 0 0 0 is redeemable after 1 0 years Coupon rate: 5 % per year, payable semi - annually X = Premium if the effective annual yield to...
-
What should be the price of a bond which has a par value of Rs . 1 0 0 0 carrying a coupon rate of 8 % and having a maturity period of 9 years? The required rate of return of the investor is 1 2 % .
-
What is the value to you of a 5 - year $ 1 , 0 0 0 face - value bond with an 1 2 % coupon rate when your yield to maturity is 9 % ?
-
You have access to two assets for investing. A Real Estate ETF, which has an expected return of 8 . 1 % , and an Agriculture Commodity ETF which has an expected return of 1 1 . 2 % . You want to...
-
Company Q s stock currently sells for $ 1 8 per share. The company just paid a dividend of $ 2 . 2 5 per share. Future dividends are expected to increase 8 % annually forever. Assume dividends are...
-
If the supply curve for fertilizer in Jordan is \(Q_{j}=-2+4 p\) and the supply curve for fertilizer in the rest of the world is \(Q_{\text {row }}=25+8 p\), what is the world supply curve?
-
You are the newly appointed tax practitioner to complete Emilys tax return and have downloaded the prefill report for Emilys tax return (hint, you can read what a prefill report is here (Links to an...
-
If 123 mL of a 1.1 M glucose solution is diluted to 500.0 mL, what is the molarity of the diluted solution?
-
Compound X, isolated from lanolin (sheep's wool fat), has the pungent aroma of dirty sweatsocks. A careful analysis showed that compound X contains 62.0% carbon and 10.4% hydrogen. No nitrogen or...
-
For each of the following structures, 1. Draw a Lewis structure; fill in any nonbonding electrons. 2. Calculate the formal charge on each atom other than hydrogen. All are electrically neutral except...
-
1. From what you remember of electronegativities, show the direction of the dipole moments of the following bonds. 2. In each case, predict whether the dipole moment is relatively large...
-
Research the different processes involved during the planning phase of a project. Identify the tasks and techniques associated with each of these processes. Prepare diagram where you relate...
-
How do cultural norms and values intersect with social structure, influencing patterns of behavior, identity formation, and societal organization ?
-
We manufacture breakfast cereal at our factory in Toad Suck, AR. Our current selling price is $2.40 per box, F.O.B. our shipping dock (our customers pay shipping expenses). Our gross profit margin is...

Study smarter with the SolutionInn App


