How would you carry out the following transformation? More than one step isneeded. CH3CH2CH2CH2C=CH T ? CCH2CH2CH2
Question:
How would you carry out the following transformation? More than one step isneeded.
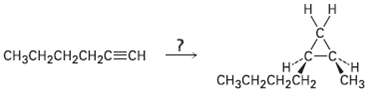
Transcribed Image Text:
нн CH3CH2CH2CH2C=CH Tн н? CнзCH2CH2CH2 СHз
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 70% (17 reviews)
Strategy The product contains a cisdisubstituted cyclopropane ring which can be formed from ...View the full answer

Answered By

Nazrin Ziad
I am a post graduate in Zoology with specialization in Entomology.I also have a Bachelor degree in Education.I posess more than 10 years of teaching as well as tutoring experience.I have done a project on histopathological analysis on alcohol treated liver of Albino Mice.
I can deal with every field under Biology from basic to advanced level.I can also guide you for your project works related to biological subjects other than tutoring.You can also seek my help for cracking competitive exams with biology as one of the subjects.
3.30+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
How would you carry out the following conversions? More than one step is needed in eachcase. "
-
How would you carry out the following reactions? More than one step may be required. (a) 3-Hexyne 3-Hexanone (b) Benzene m-Bromo acetophenone (c) Bromobenzene Acetophenone (d) 1-Methylcyclohexene ...
-
How would you carry out the following transformations? Co .CO2H (a) (b) Co CH2 C (c) CH2SH
-
Top executive officers of Preston Company, a merchandising firm, are preparing the next years budget. The controller has provided everyone with the current years projected income statement. Current...
-
Universal Technologies, Inc. has identified two qualified vendors with the capability to supply certain of its electronic components. For the coming year, Universal has estimated its volume...
-
Determine Ix in the circuit shown in Fig. 3.50 using nodal analysis. Figure 3.50 For Prob. 3.1 4 k AM 9v+
-
Stringline Ltd uses flexible budgets in order to control selling expenses. Monthly sales range from $200000 to $300000. Budgeted fixed monthly expenses for the Sales Department are $63000 for...
-
An incomplete cost of goods manufactured schedule is presented below. Instructions Complete the cost of goods manufactured schedule for MolinaCompany. MOLINA COMPANY Cost of Goods Manufactured...
-
1. Write MIPS assembly code for swapping the contents of two registers, $10 and $t1. 2. Design an algorithm for counting the number of 1's in a 32-bit number. Implement your algorithm using MIPS...
-
What are the differences between a global market segment and a national market segment? What are the marketing implications of these differences for a firm serving segments on a worldwide basis?
-
Each of the following syntheses requires more than one step. How would you carry themout? (a) CH3CH2CH2C=CH CH3CH2CH2CHO (b) (CH)2CHCH2CH CH-CH C=C (CH)2CHCH2 ?.
-
Synthesize the following compounds using 1-butyne as the only source of carbon, along with any inorganic reagents you need. More than one step may be needed. (a) 1, 1, 2, 2-Tetrachlorobutane (b) 1, 1...
-
A contract that does not satisfy the Statute of Frauds is void. True or false? Explain your answer.
-
Kerwin Industries is deciding whether to automate one phase of its production process. The manufacturing equipment has a six-year life and will cost $905,000. Projected net cash inflows are as...
-
The COVID 19 pandemic has dislodged millions of workers regionally and globally. This reality has undermined the effectiveness of the trade union movement. Critically discuss with reference to...
-
Consider the following case which illustrates a typical chain of equity investments in the startup and early growth of a venture that qualifies for venture capital: Upon incorporation, the Board...
-
Let 12 6 M == -12 -6 Find formulas for the entries of Mn, where n is a positive integer. 1 Mn -1 0 0
-
Evaluate the effectiveness of the laissez-faire leadership style present at WJAZ Radio.?
-
A: When a day of the week is randomly selected, it is a Saturday. B: When a second different day of the week is randomly selected, it is a Monday. Independent and Dependent Events. (a) determine...
-
What will be the final value of DI after executing the following piece of code? Execute the instructions dependently one after another. CLD MOU CX,OFOH MOU AX.02874H MOU DI,01000H MOU ES, DI SUB...
-
A chemist wants to make 5.5 L of a 0.300 M CaCl 2 solution. What mass of CaCl 2 (in g) should the chemist use?
-
Determine whether the following pairs of structures are actually different compounds or simply resonance forms of the same compounds. (a) (b) (c) (d) (e) (f) (g) (i) (j) and O- and and O- CH C H and...
-
Draw the important resonance forms to show the delocalization of charges in the following ions. (a) (b) (c) (d) (e) (f) (g) (h) (i) (j) CH-C CH H-C-CH CHCH CH2 NH CHCH- CH CH CH-CH 3 CH3 CH CH CH CH...
-
All of the following compounds can react as acids. Without using a table of acidities, rank them in order of increasing acidity. Explain your ranking. (a) CH3CH2SO3H (b) CH3CH2OH (c) CH3CH2COOH (d)...
-
Explain why leasing is an option for a company expansion. include, what leasing is and how it will benefit the company in it's expanding efforts. Also, how is capital or operating leasing recorded on...
-
Discuss the following statement: " A head of state signs a treaty on behalf of his country in excess of authority of his country, such treaty shal be void for inconsistency with domestic law of the...
-
A company is looking at new equipment with an installed cost of $415,329. This cost will be depreciated straight-line to zero over the project's 5-year life, at the end of which the equipment can be...

Study smarter with the SolutionInn App


