Question: Electrostatic potential maps of a typical amide (acetamide) and an acyl azide (acetyl azide) are shown. Which of the two do you think is more
Electrostatic potential maps of a typical amide (acetamide) and an acyl azide (acetyl azide) are shown. Which of the two do you think is more reactive in nucleophilic acyl substitution reactions? Explain.
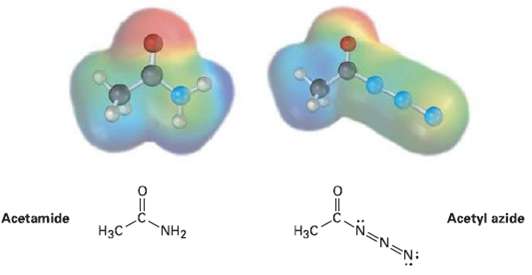
Acetamide Acetyl azide NH2
Step by Step Solution
3.44 Rating (170 Votes )
There are 3 Steps involved in it
According to the electrostatic potential maps the carbonyl carbon of ac... View full answer

Get step-by-step solutions from verified subject matter experts
Document Format (1 attachment)
22-C-O-CA (128).docx
120 KBs Word File


