Epoxides are reduced by treatment with lithium aluminum hydride to yield alcohols. Propose a mechanism for thisreaction.
Question:
Epoxides are reduced by treatment with lithium aluminum hydride to yield alcohols. Propose a mechanism for thisreaction.
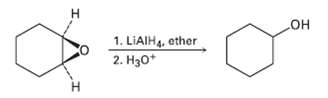
Transcribed Image Text:
н 1. LIAIH4, ether 2. Hзо* н
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 83% (12 reviews)
0 H3O 110 Protonation H Attack of hydrid...View the full answer

Answered By

Utsab mitra
I have the expertise to deliver these subjects to college and higher-level students. The services would involve only solving assignments, homework help, and others.
I have experience in delivering these subjects for the last 6 years on a freelancing basis in different companies around the globe. I am CMA certified and CGMA UK. I have professional experience of 18 years in the industry involved in the manufacturing company and IT implementation experience of over 12 years.
I have delivered this help to students effortlessly, which is essential to give the students a good grade in their studies.
3.50+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Epoxide?s react with Grignard reagents to yield alcohols propose a mechanism. 1. CHMgBr 2. * CH
-
Propose a mechanism for each reaction. (a) (b) (c) (d) OH H2SO heat OCH + CH3OH H20 CH2OH H2So4 heat CH OCH2CH CH CH,CH2OH (a minor product)
-
Propose a mechanism for each reaction. (a) (b) Ht H3C CH3 OH H SOA Ph Ph Ph Ph
-
When considering banning a toxic chemical, why should replacement substances or processes be part of the decision?
-
What are the big differences in the way communities approach development today compared to 1990, when Porter wrote The Competitive Advantage of Nations?
-
Determine the standard molar entropy of H 35 Cl at 298 K, where B = 10.58 cm 1 , = 2886 cm 1 , the ground-state electronic level degeneracy is 1, and P = 1.00 atm.
-
Culture and Mental Illness A recent study examining the link between schizophrenia and culture interviewed 60 people who had been diagnosed with schizophrenia and who heard voices in their heads. The...
-
Mobility Partners makes wheelchairs and other assistive devices. For years it has made the rear wheel assembly for its wheelchairs. A local bicycle manufacturing firm, Trailblazers, Inc., offered to...
-
Salvador Industries bought land and built its plant 20 years ago. The depreciation on the building is calculated using the straight-line method, with a life of 30 years and a salvage value of $52.000...
-
Hospital personnel routinely examine patient records for error, such as incomplete insurance information, on incomplete patient history, or missing/incomplete medical records. On average, about 250...
-
Safrole, a substance isolated from oil of sassafras, is used as a perfumery agent. Propose a synthesis of safrole from catechol (1,2-benzenediol). CH2CH=CH2 Safrole
-
Show the structure and stereochemistry of the alcohol that would result if 1, 2-epoxycyclohexane (Problem 18.39) were reduced with lithium aluminum deuteride, LiAID4.
-
The Federal Reserve exercises monetary policy by means of a very short-term, overnight nominal interest rate. Explain how changes in this overnight nominal rate influence longer term real interest...
-
You are a financial accountant working at Knappa Foods Limited ( Knappa ) . Knappa is a manufacturer and retailer of food products. It has two manufacturing facilities in Australia one in Gosford,...
-
Simon LLC produces smartphones, computers, and televisions. Although all high - level decisions are made at company headquarters by top management, rapid expansion and increasingly specialized...
-
What component is not a part of budgeted cost sheet?
-
At the end of the current year, the company had total assets of $7,600. During the year, the company recorded sales of $3,900 and expenses of $2,200. Its board of directors declared $500 in dividends...
-
UXZ is a wedding event organiser company based in Z - land. The Finance Manager at UXZ would like to use activity - based costing principles to apply the correct overhead costs to the three wedding...
-
What limitations do courts impose on police in the use of deception in interrogations of suspects?
-
Refer to the data in QS 10-1. Based on financial considerations alone, should Helix accept this order at the special price? Explain.
-
The Ocean Thermal Energy Conversion (OTEC) project in Hawaii produces electricity from the temperature difference between water near the surface of the ocean (about 27 C) and the 600 m deep water at...
-
Write resonance structures for the azide ion, N3-. Explain how these resonance structures account for the fact that both bonds of the azide ion have the same length.
-
Write structural formulas of the type indicated: (a) Bond-line formulas for seven constitutional isomers with the formula C4H10O; (b) Condensed structural formulas for two constitutional isomers with...
-
Write the Lewis structure of (a) CH2Fl2 (difluoromethane) (b) CHCl3 (chloroform).
-
A representative household maximizes. where . BlogC1+i) j=0 Ct+j+kt+j (3) (4) Note that the second term of the right-hand side of the equation is expressed by not Kt+j-1 but Kt+j-2. In other words,...
-
Two parallel plates 15 cm on a side are given equal and opposite charges of magnitude 5.5 x 10-9 C. The plates are 3.5 mm apart. What is the potential difference between the plates (in V)? (Enter the...
-
The equipotential lines in a region of electric field are shown in the diagram below. For each path indicated below, what is the work do by the electric field in moving a charge q = +7.5 x 107 C...

Study smarter with the SolutionInn App


