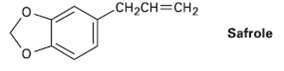
Safrole, a substance isolated from oil of sassafras, is used as a perfumery agent. Propose a synthesis
Question:
Safrole, a substance isolated from oil of sassafras, is used as a perfumery agent. Propose a synthesis of safrole from catechol (1,2-benzenediol).
Transcribed Image Text:
CH2CH=CH2 Safrole
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (8 reviews)
OH OH 2 NaH O ...View the full answer

Answered By

Shyamsunder K
Teaching my kids and students ( though I am not a teacher by profession) around me and helping them in easily comprehend the subjects; though teaching is my passion but not as profession and students who I have taught have achieved success in taking the exams confidently and perform great;y.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Karahanaenone, a terpenoid isolated from oil of hops, has been synthesized by the thermal reaction shown. Identify the kind of pericyclic reaction, and explain how karahanaenone isformed. CH CH eat...
-
Safrole is contained in oil of sassafras and was once used to flavor root beer. A 2.39-mg sample of safrole was dissolved in 103.0 mg of diphenyl ether. The solution had a melting point of 25.70C....
-
Propose a formula for hydrogen peroxide, a substance used as a bleaching agent. (Curiously, this compound does not behave as an acid, despite its formula. It behaves more like a classic...
-
The following financial statements for Brownstone plc are a slightly simplified set of published accounts. Brownstone plc is an engineering business that developed a new range of products in 2007....
-
Evaluate how Apple can gain business intelligence through the implementation of a customer relationship management system.
-
How would you expect the internal energy of 79 BrF to compare to that of 79 Br 35 Cl at 298 K? Check your answer by using the following data: V (cm-1) B (cm-1) 19BIF 671 0.356 79 Br3SC1 0.153 420
-
Help for Insomniacs In Exercise 1.23, we learned of a study to determine whether just one session of cognitive behavioral therapy can help people with insomnia. In the study, forty people who had...
-
The following are brief descriptions of several companies in different lines of business. A. Company A is a construction company. It has recently signed a contract to build a highway over a...
-
On January 1, 2019, the partner's capital balance in Rosen & Decker Ltd are Jerry Rosen, $27,000 and Patricia Decker, $34,000. For the year ended December 31, 2019, the partnership reports a profit...
-
What are two significant ways slavery has determined the self-perceptions of the Haitian people; try to consider masters, slaves, and others (such as immigrants, foreign business agents, and other...
-
Meerwein?s reagent, triethyloxonium tetra-fluoroborate, is a powerful ethylating agent that converts alcohols into ethyl ethers at neutral pH. Show the reaction of Meerwein?s reagent with...
-
Epoxides are reduced by treatment with lithium aluminum hydride to yield alcohols. Propose a mechanism for thisreaction. 1. LIAIH4, ether 2. H*
-
A unique identifier for a data row that consists of more than one field is commonly called a: a. Primary plus key b. Composite primary key c. Foreign key d. None of the above
-
What are the six factors courts may use to determine if the claimed damages are reasonably certain?
-
1.Explain why it is more difficult to work out the contribution per unit of different pieces of furniture produced by one carpenter compared to the contribution per unit of the output of a large high...
-
1 . Please explain sustainability reporting 2 . What is the major challenge ( s ) a company faces in preparing the sustainability reporting made public? 3 . What result ( s ) or conclusion ( s ) may...
-
If a non domiciled non citizen died owning $ 1 . 5 million of US situs assets, what is the estate tax?
-
One of the vesting schedules for ERISA provides for full vesting of the employer's contribution to an employee's pension fund after five years or gradually over, Please describe what is the actual...
-
What factors do courts consider in determining whether an individual is in custody when a police interrogation takes place?
-
On the basis of the details of the following fixed asset account, indicate the items to be reported on the statement of cashflows: ACCOUNT Land ACCOUNT NO. Balance Date Item Debit Credit Debit Credit...
-
A tank contains 20 percent liquid water and 80 percent steam by volume at 200 C. Steam is withdrawn from the top of the tank until the fluid remaining in the tank is at a temperature of 150 C....
-
Consider another chemical species like the ones in the previous problems in which a carbon atom forms three single bonds to three hydrogen atoms but in which the carbon atom possesses a single...
-
Draw a three-dimensional orbital representation for each of the following molecules, indicate whether each bond in it is a s or p bond, and provide the hybridization for each non-hydrogen atom. (a)...
-
Ozone (O3) is found in the upper atmosphere where it absorbs highly energetic ultraviolet (UV) radiation and thereby provides the surface of Earth with a protective screen. One possible resonance...
-
How do I use voice commands to control my computer with the project of making a video?
-
Dan Mullen is employed by a mail-order distributor and reconditions used desktop computers, broadband routers, and laser printers. Dan is paid $12 per hour, plus an extra $6 per hour for work in...
-
Consider two series Yt and Y2t: Y = IIY-1 +6 Or more explicitly as: Aye Aya Yit Elt + Y2t 2t in which = [1, 2]' is IID with E(e) = 0 and E(ee'): [ 0 0 a) Write the system in levels Answer: A-AU-N b)...

Study smarter with the SolutionInn App


