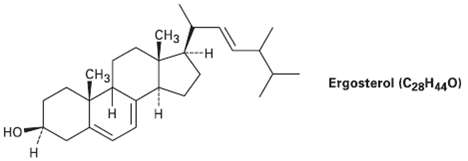
Ergo sterol a precursor of vitamin D, has ? max = 282 nm and molar absorptivity ?
Question:
Ergo sterol a precursor of vitamin D, has ? max = 282 nm and molar absorptivity ? = 11,900. What is the concentration of ergo sterol in a solution whose absorbance A = 0.065 with a sample path length l = 1 .00cm?
Transcribed Image Text:
сНз Ergosterol (C28H440) снз но I.
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 72% (11 reviews)
c A ex 0065 1...View the full answer

Answered By

AJIN kuriakose
I have completed B.Tech in Electrical Engineering & Masters in Power & Control From one of the best universities in India. I got the 99.05 percentile in the Gate Electrical Engineering Exam. I can Help students solving assignments in Electrical subjects like Power Electronics, Control system, Analog, Network Theory & Engineering Mathematics. Clear your fundamentals and develop problem-solving skills and analytical skills to crack the exam.
Get guidance and the opportunity to learn from experienced...
I can provide tuition for Electrical engineering subjects (Power Electronics, Digital electronics, Network Theory, Control System & Engineering Mathematics). The toughest subject of Electrical engineering can be made simple in online classes...
I can also solve it.
1 .I can help you with your assignments or exams or quiz or tutoring.
2. Very strict to the deadlines.
Message me for any help in assignments, live sessions. I am here to help students for all assignments, tests and exams and I will make sure you always get _95% In your subject.
Contact me in solution inn for any help in your semester, projects and for many more things . Also feel free to contact me through solution inn and for any advise related to tutoring and how it works here.thank you.
5.00+
5+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
What is the concentration of hydroxide ion in a 0.060 M aqueous solution of methylamine, CH3NH2? What is the pH?
-
What is the concentration of hydroxide ion in a 0.18 M aqueous solution of hydroxylamine, NH2OH? What is the pH?
-
What is the concentration of hydroxide ion in a 0.21 M aqueous solution of hydroxylamine, NH 2 OH? What is the pH?
-
Sandys Socks makes the worlds best socks. Information for the last eight months follows: Prepare a scatter graph by plotting Sandys data on a graph. Then draw a line that you believe best fits the...
-
If you wanted to hire an accountant to work in New York City, would you search the database for wages in the New York City metropolitan area or for the whole state? Why?
-
(a) Write the anode and cathode reactions that cause the corrosion of iron metal to aqueous iron(II). (b) Write the balanced half-reactions involved in the air oxidation of Fe2+ (aq) to Fe2O3 3 H2 O.
-
In performing analytical procedures in the expenditure/disbursement cycle, an auditor detects that payables divided by current liabilities appears low in comparison with prior years. Which of the...
-
Presented below are selected transactions at Ridge Company for 2015. Jan. 1 Retired a piece of machinery that was purchased on January 1, 2005. The machine cost $62,000 on that date. It had a useful...
-
A solid steel shaft is 14 ft. long has a diameter of 6 inches over 9 ft. of its length and a diameter of 4 inches for the remaining length, as shown The shaft is in equilibrium when subjected to the...
-
The entity reports the following transactions for the 2022 tax year. The trustee accumulates all accounting income for the year. Operating income from a business $500,000 30,000 Dividend income, all...
-
Adiponitrile, a starting material used in the manufacture of nylon, can be prepared in three steps from 1, 3-butadiene. How would you carry out thissynthesis? 3 steps H2C=CHCH=CH2 N=CCH2CH2CH2CH2C=N...
-
A 1, 3-Cyclopentadiene polymerizes slowly at room temperature to yield a polymer that has no double bonds except on the ends. On heating, the polymer breaks down to regenerate 1, 3-cyclopentadiene....
-
The composite shaft shown in the figure is manufactured by shrink-fitting a steel sleeve over a brass core so that the two parts act as a single solid bar in torsion. The outer diameters of the two...
-
Microsoft has a bond that was issued two years ago. It has a par value of $3,000, the coupon rate of 9%, paid semiannually , and has 4 more years left until it matures. Its yield to maturity is 10%....
-
Assume a world without inflation. Janice Lewis berg, 38, plans to retire at age 68. Her life expectancy is age 90. She wants to live a retirement lifestyle that will cost $35,000 per year, payable at...
-
Mark's yearly salary is $89,000 and he deposits 25% of it into his savings account at the end of every year. The interest rate the bank offers is 3.1% per year. It has been 3 years since Mark started...
-
How do operating systems implement security measures for inter-process communication (IPC), and what are the potential security risks associated with IPC mechanisms like shared memory and message...
-
Evaluate the concept of endpoint detection and response (EDR) in the context of operating system security. What are the key components of an EDR solution, and how does it differ from traditional...
-
During the \(0.75 \mathrm{~s}\) that the tube is running, what is the electric power? A. \(7.0 \mathrm{~kW}\) B. \(700 \mathrm{~W}\) C. \(70 \mathrm{~W}\) D. \(7.0 \mathrm{~W}\) \(\mathrm{X}\) rays...
-
Provide an example of an aggressive accounting practice. Why is this practice aggressive?
-
The enthalpy of solution for cesium fluoride is -36.8 kJ/mol. What can you conclude about the relative magnitudes of Hsolute and Hhydration ? (a) AHsolute>|AHhydration (b) AHsolute
-
Determine which of the heterocyclic amines just shown are aromatic. Give the reasons for your conclusions.
-
(a) Show how fragmentation occurs to give the base peak at m/z 58 in the mass spectrum of ethyl propyl amine (N-ethylpropan-1- amine), shown below. (b) Show how a similar cleavage in the ethyl group...
-
Propose a mechanism for nitration of pyridine at the 4-position, and show why this orientation is not observed.
-
Schwiesow Corporation has provided the following information: Cost per Cost per Unit Period Direct materials $ 7.05 Direct labor $ 3.50 Variable manufacturing overhead $ 1.65 Fixed manufacturing...
-
Choose an industry in which you are interested in working. How is that industry trending? What internal and external factors may affect the direction of the organization? For your initial post,...
-
The DeVille Company reported pretax accounting Income on its Income statement as follows: 2024 2025 2026 2027 $ 420,000 340,000 410,000 450,000 Included in the income of 2024 was an installment sale...

Study smarter with the SolutionInn App


