Ethyl dimethylacetoacetate reacts instantly at room temperature when treated with ethoxide ion to yield two products, ethyl
Question:
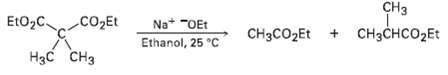
Ethyl dimethylacetoacetate reacts instantly at room temperature when treated with ethoxide ion to yield two products, ethyl acetate and ethyl 2-mcthylpropanoatc. Propose a mechanism for this cleavagereaction.
Transcribed Image Text:
CHз CH3CHCO2ET EtO,C. CO2Et Na* "OEt Ethanol, 25 °C CH3CO2ET + Нзс Cнз
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (8 reviews)
Go CHCCCH32COEtCHCCCHCOEt addition of ...View the full answer

Answered By

PALASH JHANWAR
I am a Chartered Accountant with AIR 45 in CA - IPCC. I am a Merit Holder ( B.Com ). The following is my educational details.
PLEASE ACCESS MY RESUME FROM THE FOLLOWING LINK: https://drive.google.com/file/d/1hYR1uch-ff6MRC_cDB07K6VqY9kQ3SFL/view?usp=sharing
3.80+
3+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Propose a mechanism for each reaction. (a) (b) (c) (d) OH H2SO heat OCH + CH3OH H20 CH2OH H2So4 heat CH OCH2CH CH CH,CH2OH (a minor product)
-
Propose a mechanism for each reaction. (a) (b) Ht H3C CH3 OH H SOA Ph Ph Ph Ph
-
Propose a mechanism for the acid-catalyzed condensation of n-propyl alcohol to n-propyl ether, as shown above. When the temperature is allowed to rise too high, propene is formed. Propose a mechanism...
-
Two positive charges, each with charge q = 2.5 nC, are placed as shown in the diagram. The distance d 0.42 m. Find the net electric potential at the point x = 0, y = 0.12 m.
-
Consider an industry in which firms can expect to sell 1000 units annually at a price of P. Before firms enter, they do not know their production costs with certainty. Instead, they believe that unit...
-
The National Institute of Mental Health reports that there is a 20% chance of an adult American suffering from a psychiatric disorder. Four randomly selected adult Americans are examined for...
-
Describe the role of the patient, physician, nurse, and hospital in obtaining informed consent.
-
Marble Construction estimates that its WACC is 10% if equity comes from retained earnings. However, if the company issues new stock to raise new equity, it estimates that its WACC will rise to 10.8%....
-
mework #3 i Saved Check my work mode: This shows what is correct or incorrect for the work you have comp The year-end financial statements of Cobra Tax Services are provided below. Service revenue...
-
The Wilcox Student Health Center has just implemented a new computer system and service process to improve efficiency. The process flowchart and analysis framework is also provided. As pharmacy...
-
Give the structures of the possible Claisen condensation products from the following reactions. Tell which, if any, you would expect to predominate in each case. (a) CH3CO2Et + CH3CH2CO2Et (b)...
-
In contrast to the rapid reaction shown in Problem 23.40, ethyl acetoacetate requires a temperature over 150 ?C to undergo the same kind of cleavage reaction. How can you explain the difference in...
-
The student loan association raises floating-rate financing and makes loans of maturity 510 years at fixed rates. Can you describe the nature of the risks on the balance sheet? What financial...
-
how do i calculate Price-to-Book (P/B) Price-to-Earnings (P/E) Price-to-Revenue (P/R)
-
Compost Inc. (CI) has a 8% return on equity, a reinvestment ratio of 70% and is expected to pay a $7 dividend. Suppose that the business is growing at 2% per year, and the price per share is $180....
-
In a cost matrix, what does the cost of a risk refer to? Question 12Answer a. The probability of the risk occurring b. The severity of the risk c. The duration of the risk d. The estimated cost of...
-
Commodity markets are mark-to-market daily. Question 9 options: True False
-
In chapter 9, is Risk Management mandated within the federal laws governing college/universities? Question 10 options: A) Yes, by the Higher Education Administrative and Regulatory body that performs...
-
Maria is a single individual with taxable income of $75,000 in 2017. What marginal tax rate should she use to determine the tax savings from a $2,000 deductible expense?
-
Willingness to pay as a measure of a person's value for a particular good measures the maximum a person would be willing to pay requires that payment actually be made depends on the satisfaction that...
-
Write the Lewis structure for each molecule or ion. a. H 3 COCH 3 b. CN c. NO 2 d. ClO
-
Give a correct name for each compound. (a) (b) (c) (d) (e) (f) CH CH3CH22C CHCH Cl CH,CI
-
(a) Draw and name all five isomers of formula C3H5F. (b) Draw all 12 acyclic (no rings) isomers of formula C4H7Br. Include stereoisomers. (c) Cholesterol, C27H46O, has only one pi bond. With no...
-
Draw and name all stereoisomers of 3-chlorohepta-2,4-diene (a) Using the cis-trans nomenclature. (b) Using the E-Z nomenclature.
-
Ramirez Company installs a computerized manufacturing machine in its factory at the beginning of the year at a cost of $45,300. The machine's useful life is estimated at 10 years, or 403,000 units of...
-
14. (3 points) Write a program that ask the user for 1. their first name and 2. their last name, Enter first name: Matt Enter last name: Priem Hello Matt Priem! and then outputs a greeting similar to...
-
Given a sorted array 2, 5, 8, 12, 16, 18, 22, 25, 29, 32 and the following interpolation search algorithm. Show the steps of the algorithm when 25 is searched. Also, when 26 is searched. array a,...

Study smarter with the SolutionInn App


