Question: Explain how UV spectroscopy could be used to distinguish between thesecompounds. and a) b) and c) and HO and d)
Explain how UV spectroscopy could be used to distinguish between thesecompounds.
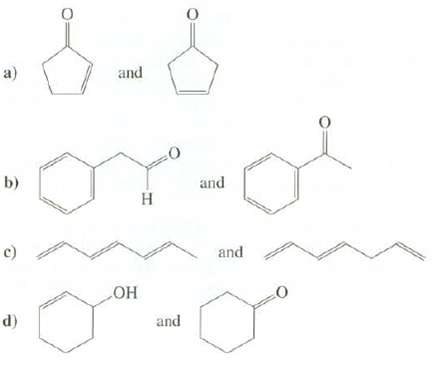
and a) b) and c) and HO and d)
Step by Step Solution
3.42 Rating (168 Votes )
There are 3 Steps involved in it
a The ketone on the left is conjugated and should absorb ... View full answer

Get step-by-step solutions from verified subject matter experts
Document Format (1 attachment)
15-C-O-S (100).docx
120 KBs Word File


