Explain the difference in the melting points of these isomers: mp=-140C 0 mp = 7C
Question:
Explain the difference in the melting points of these isomers:
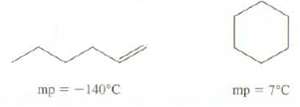
Transcribed Image Text:
mp=-140°C 0 mp = 7°C
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 65% (20 reviews)
The cycloalkane has a hig...View the full answer

Answered By

Utsab mitra
I have the expertise to deliver these subjects to college and higher-level students. The services would involve only solving assignments, homework help, and others.
I have experience in delivering these subjects for the last 6 years on a freelancing basis in different companies around the globe. I am CMA certified and CGMA UK. I have professional experience of 18 years in the industry involved in the manufacturing company and IT implementation experience of over 12 years.
I have delivered this help to students effortlessly, which is essential to give the students a good grade in their studies.
3.50+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Explain the difference in the melting points of the following compounds: Only one of the two can form intramolecular hydrogen bonds? NO2 NO OH OH m.p. 115C m.p. 45C
-
Explain the difference in the melting points of the following fatty acids: a. Palmitic acid and stearic acid b. Palmitic acid and palmitoleic acid c. Oleic acid and linoleic acid
-
Explain the difference in boiling points between 1-butanol and ethanol in terms of the intermolecular forces.
-
____, one-way ANOVA compares the means of three or more independent samples.
-
Fletcher Industries manufactures a range of household products. Roger Wilkie is head of the Hardware Division and he has just experienced a terrible nine months. 'If it could go wrong it did. Sales...
-
Roger Ginde is developing a program in supply chain management certification for managers. Ginde has listed a number of activities that must be completed before a training program of this nature...
-
How does multiplicity relate to business rules? Give an example.
-
(a) Plot the above data on a graph. Do you observe any trend, cycles, or random variations? (b) Starting in year 4 and going to year 12, forecast demand using a 3-year moving average. Plot your...
-
The Rolling Department of Jabari Steel Company had 5,300 tons in beginning work in process inventory (70% complete) on October 1. During October, 87,800 tons were completed. The ending work in...
-
1. To what degree do you think Casper has disrupted the mattress industry? To what degree did Caspers research about the mattress buying process resonate with you? Would you buy a Casper mattress?...
-
Explain the differences in the boiling points between the members of each of these pairs of compounds: (a) CH 3 (CH 2 )6 CH3 bp: 126C CH3 (CH2)8 CH3 bp: 174C (b) CH3CH2CH2OH bp: 97C CH3CH2OCH3 bp:...
-
Explain which compound you expect to have the higher melting point. c) of b) CH(CH)1CH or or CH(CH)29CH, Beeswax
-
Find the lengths of the limacons r = 2 + cos and r = 2 + 4 cos .
-
A piston-cylinder assembly contains \(3 \mathrm{~kg}\) of air at 15 bar and \(620 \mathrm{~K}\). The environment is at a pressure of 1 bar and \(300 \mathrm{~K}\). The air is expanded in a fully...
-
Show \[\begin{aligned} \text { (a) } T \mathrm{~d} s & =c_{p} \mathrm{~d} T-T\left(\frac{\partial v}{\partial T} ight)_{p} \mathrm{~d} p \\ \text { (b) } T \mathrm{~d} s & =c_{v}\left(\frac{\partial...
-
Show that if the ratio of the specific heats is 1.4 , then \[\left(\frac{\partial p}{\partial T} ight)_{s}=\frac{7}{2}\left(\frac{\partial p}{\partial T} ight)_{v}\]
-
An air-standard Diesel cycle operates with a compression ratio, \(r=10: 1\). If the initial conditions at bdc are \(1 \mathrm{bar}\) and \(27^{\circ} \mathrm{C}\), and the energy addition is \(2000...
-
For a van der Waals gas that obeys the state equation \[p=\frac{R T}{v-\mathrm{b}}-\frac{\mathrm{a}}{v^{2}}\] shows that the coefficient of thermal expansion, \(\beta\), is given by \[\beta=\frac{R...
-
Aaron Feuerstein was the third generation chief executive officer and chairman of the board of Malden Mills, a privately held company started in Massachusetts that produced fabric and evolved to...
-
Walker, Inc., is an all-equity firm. The cost of the company's equity is currently 11.4 percent and the risk-free.rate is 3.3 percent. The company is currently considering a project that will cost...
-
(a) Evaluate the Riemann sum for f(x) = x 3 6x taking the sample points to be right endpoints and a = 0, b = 3, and n = 6. (b) Evaluate (x - (x - 6x) dx.
-
When 2-methyl-2, 5-pentanediol is treated with sulfuric acid, dehydration occurs and 2, 2-dimethyltetrahydrofuran is formed. Suggest a mechanism for this reaction. Which of the two oxygen atoms is...
-
Write the mechanism of the hydrolysis of cis-5, 6-epoxydecane by reaction with aqueous acid. What is the stereochemistry of the product, assuming normal backside SN2 attack?
-
What is the stereochemistry of the product from acid-catalyzed hydrolysis of trans-5, 6-epoxvdecane? How does the product differ from that formed in Problem 18.33?
-
Write an equation for the polynomial graphed below 5+ 4 + -5-4-3-2 y = 3. 2- 1 + + 2 3 4 5 -2 345 -5+
-
How can ethical leadership principles be leveraged to navigate the complexities of organizational dynamics and promote a culture of integrity, accountability, and social responsibility ?
-
Consider the 12-QAM constellation shown in Figure 2 and the baseband pulse, g(t) depicted in Figure 3. The 12 passband transmit waveforms are represented as Sm1(t) = Amig(t) cos (2fet) - Amag(t) sin...

Study smarter with the SolutionInn App


