Explain whether these reactions follow an SN1 or an SN2 mechanism. CH3 T a) CH3-C-Br + CHCO
Question:
Explain whether these reactions follow an SN1 or an SN2 mechanism.
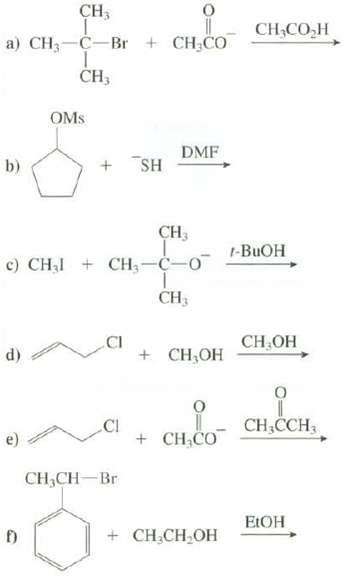
Transcribed Image Text:
CH3 T a) CH3-C-Br + CH₂CO CH3 b) OMS d) + SH CH3 c) CH₂ CH₂-C-0 T CH3 DMF CH₂CH-Br + CH₂OH + CH CO + CH₂CH₂OH CH,COH 1-BUOH CH₂OH CH₂CCH EtOH
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (8 reviews)
a The substrate is a tertiary alkyl halide so the reaction follows the SN1 mechan...View the full answer

Answered By

Robert Mwendwa Nzinga
I am a professional accountant with diverse skills in different fields. I am a great academic writer and article writer. I also possess skills in website development and app development. I have over the years amassed skills in project writing, business planning, human resource administration and tutoring in all business related courses.
4.90+
187+ Reviews
378+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
In contrast to SN2 reactions, SN1 reactions show relatively little nucleophile selectivity. That is, when more than one nucleophile is present in the reaction medium, SN1 reactions show only a slight...
-
Explain whether this reaction would follow the SN1 or the SN2 mechanism and then explain which reaction is faster: CI a) CHCH + OH HO CHOH CH3 b) CHC-Br + CHCO CH, O-SOCH3 or CHCH CH CO,H or + OH CH3...
-
Explain the difference between an SN1 reaction mechanism and an SN2 reaction mechanism.
-
Explain the economic basis for the U shape of the long-run average total cost curve.
-
Leather Products Ltd manufactures leather goods. The company's profits have declined during the past nine months. In an attempt to isolate the causes of poor profit performance, management is...
-
Paul owns all 100 shares of Presto Corporation stock. His basis in the stock is $10,000. Presto has $100,000 of E&P. Presto redeems 25 of Pauls shares for $30,000. What are the tax consequences of...
-
In Example 12.3, how will the maximum height, \(z_{1}\), that the pump can be located above the water surface change if the water temperature is decreased to \(40^{\circ} \mathrm{F}\)? Example 12.3 A...
-
The following data are for four independent process-costing departments. Inputs are added continuously. Required: Compute the equivalent units of production for each of the preceding departments...
-
Determine the cash fixed costs which are used when calculating EBDAT: Administrative expenses = $ 1 0 0 , 0 0 0 ; Rent expenses = $ 7 0 , 0 0 0 ; Depreciation expenses = $ 5 0 , 0 0 0 ; and Interest...
-
Based in central London the European Club was founded in 1900. The club is a non-profit making charity whose good works for the last 100+ years include offering scholarships for gifted children from...
-
Explain in which solvent these reaction are faster: a) HC Br in CHOH or CH-CHOH I b) CHCHCHCH + OH in CHOH or 50% CHOH 50% HO CHICHICH c) CHCHCH + C=N: in CHCHOH or DMSO
-
Show the products of these reactions. (Remember that acid-base reactions are usually much faster than nucleophilic substitution reactions.) a) C1-CHCHCHCOH b) Br OH NH3 + OH
-
For Problems use the definition of the derivative to obtain the following results. If f(x) = x 4 , then f'(x) = 4x 3 .
-
For Problem 8.14, calculate the real and reactive power delivered to the three-phase load. Data From Problem 8.14:- The voltages given in Problem 8.10 are applied to a balanced-Y load consisting of...
-
Rework Problem 6.2 with the value of \(\mathrm{A}_{11}\) changed to 4 . Data From Problem 6.2:- Using Gauss elimination and back substitution, solve \[\left[\begin{array}{ccc}6 & 2 & 1 \\4 & 10 & 2...
-
Repeat the above problem with the swing bus voltage changed to \(1.0 / 30^{\circ}\) per unit. Data From Above Problem:- Assume a \(0.8+j 0.4\) per unit load at bus 2 is being supplied by a generator...
-
The interest expense on debt provides a tax deduction for the issuer so any calculation of a firms net cost of debt should reflect this benefit. Based on the information provided at MFL, compute a...
-
Repeat Problem 6.37 except use an initial voltage guess of \(0.25 / 0^{\circ}\). Data From Problem 6.37:- The bus admittance matrix of a three-bus power system is given by \[ Y_{\text {bus...
-
Find the indicated critical value. z 0.03
-
Ashlee, Hiroki, Kate, and Albee LLC each own a 25 percent interest in Tally Industries LLC, which generates annual gross receipts of over $10 million. Ashlee, Hiroki, and Kate manage the business,...
-
Solve each system by graphing. 1 3 -2 1' p=3x+3 17 -3 -2 -1 4 m N H let HA f Her
-
Identify the reagents a?c in the following scheme: H.
-
Organo metallic reagents such as sodium acetylide undergo an addition reaction with ketones, giving alcohols: How might you use this reaction to prepare 2-methyl-1, 3-butadiene, the starting material...
-
The oral contraceptive agent Mestranol is synthesized using a carbonyl addition reaction like that shown in Problem 8.42. Draw the structure of the ketoneneeded. CH3 CECH Mestranol CH30
-
ow do synergistic dynamics emerge within high-performance teams, and what role do diverse skill sets, complementary roles, and shared goals play in fostering collaborative innovation and collective...
-
Pietro Frozen Foods, Inc., produces frozen pizzas. For next year, Pietro predicts that 50,000 units will be produced, with the following total costs: Direct materials Direct labor Variable overhead...
-
You will complete two case studies over the course of the semester. These are to be completed individually. Each case study is found in the textbook and there are questions there to help guide your...

Study smarter with the SolutionInn App


