Question: Explain whether this reaction would follow the SN1 or the SN2 mechanism and then explain which reaction is faster: CI a) CHCH + OH HO
Explain whether this reaction would follow the SN1 or the SN2 mechanism and then explain which reaction is faster:
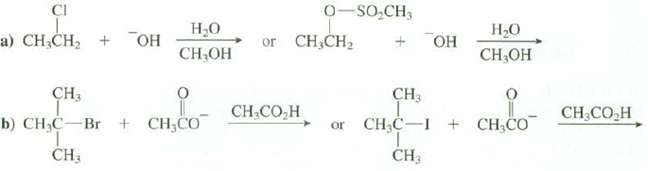
CI a) CHCH + OH HO CHOH CH3 b) CHC-Br + CHCO CH, O-SOCH3 or CHCH CH CO,H or + OH CH3 CH3 HO CH,OH + CH CO CH3COH
Step by Step Solution
3.45 Rating (168 Votes )
There are 3 Steps involved in it
a Primary substrates with a strong nucleophile hydroxide ion reac... View full answer

Get step-by-step solutions from verified subject matter experts
Document Format (1 attachment)
15-C-O-N-R (9).docx
120 KBs Word File


