Explain which compound has a faster rate of SN1 reaction. a) c) HC CI CI or or
Question:
Explain which compound has a faster rate of SN1 reaction.
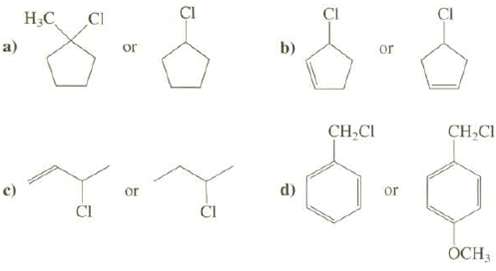
Transcribed Image Text:
a) c) H₂C CI CI or or J d) CI CH₂CI or or D CH₂Cl OCH 3
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 54% (11 reviews)
For the S N 1 reaction formation of the carbocation is the rate limitin...View the full answer

Answered By

Shaira grace
I have experience of more than ten years in handing academic tasks and assisting students to handle academic challenges. My level of education and expertise allows me communicate eloquently with clients and therefore understanding their nature and solving it successfully.
5.00+
4+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Explain which compound has a faster rate of reaction withHCI: b) or or NO2 or
-
Explain which compound has a faster rate of SN2 reaction: a) HC CI CI c) PhCHCH3 or H CI Cl or CHCHCH3 b) CI CH3 CH3 or U
-
Which of these compounds would have faster rate of SN1 reaction? a) b) c) d) e) CI ta Ph Ph -CI or Br or Br or or -Cl or + CH3 Ph -Cl Br Br ta -CI
-
What other types of contingency planning should Matt and Chris include to make the report comprehensive? Please explain the relevance of each suggestion.
-
Describe the problems that can arise in using process costing to estimate the cost of the services produced in mass service entities.
-
On December 30, 2013, Maud sold land to her son, Charles, for $50,000 cash and a 7% installment note for $350,000, payable over 10 years. Maud's cost of the land was $150,000. In October 2015, after...
-
What is the future equivalent of \($1,000\) invested at 8% simple interest for 3 years?
-
Howell made long-distance telephone calls through the telephone companys computer-controlled switching system to solicit funding for a nonexistent business enterprise. What crimes did Howell commit?...
-
9. Write a trinomial of the form x + bx+c that is prime.
-
The rod assembly is used to support the 250-lb cylinder. Determine the components of reaction at the ball-and socket joint A, the smooth journal bearing E, and the force developed along rod CD. The...
-
Consider the free energy versus reaction progress diagram for the SN2 reaction shown in Figure 8.1. Does the transition state for this reaction have the C Cl bond less than half broke, approximately...
-
Arrange these compounds in order of decreasing SN1 reaction rate. Ph CI CI CI Ph CI Ph
-
What are the causes and symptoms of Addison disease and Cushing syndrome?
-
Using the St. Louis Fed website (FRED), chart the Merrill Lynch HighYield (speculative grade) Master II optionadjusted corporate bond spread. How has it behaved over the business cycle?
-
Prepare a flowchart on obtaining an injunction (or restraining order) against violence threatened by a husband against his wife in your state.
-
Media Moguls hired Charissa Love, an African-American, for the position of social media coordinator. Revise the following sentences to reduce bias (e.g., gender, racial, ethnic, age, and disability).
-
An ideal gas initially at volume \(V_{1}\), pressure \(P_{1}\), and temperature \(T_{1}\) undergoes an isobaric process that changes its temperature to \(T_{2}\). The gas immediately undergoes an...
-
Graphically depict a shut-down case for a monopolistic competitive firm. When should any firm shut down in the short run?
-
The pulley of mass \(M\) shown in Figure 5.126 has a radius of \(r\). The mass moment of inertia of the pulley about the point \(\mathrm{O}\) is \(I_{\mathrm{O}}\). A translational spring of...
-
Tanaka Company's cost and production data for two recent months included the following: March April Production (units).........300................600 Rent.....................$1,800............$1,800...
-
Find the slope of the line through each pair of points. (8,9), (8, 16)
-
How would you prepare ds-2-butene starting from propyne, an alkyl halide, and any other reagents needed? This problem cant be worked in a single step. Youll have to carry out more than one reaction.
-
Beginning with 4-octyne as your only source of carbon, and using any inorganic reagents necessary, how would you synthesize the following compounds? (a) cis-4-Octene (b) Butanal (c) 4-Bromooctane (d)...
-
Beginning with acetylene and any alkyl halides needed, how would you synthesize the following compounds? (a) Decane (b) 2, 2-Dimethylhexane (c) Hexanal (d) 2-Heptanone
-
United States Dominance in Psychology Collaboration is a critical feature of global psychology. In order for professionals to work together throughout the world, it is important for them to have some...
-
( support your answer with one academic reference and non-academic or industry reference ). Every year, as a CEO of a company, you are required to make an annual formal report. In your opinion, what...
-
Question 1 Apple is back under the spotlight over labor conditions in its supply chain following an explosive report from The Information on Thursday that revealed new details about the company's...

Study smarter with the SolutionInn App


