Question: Explain which functional group(s) is present in the compound that has this IRspectrum: 80 60 40 20 1721 cm 3500 4000 3000 2500 1500 2000
Explain which functional group(s) is present in the compound that has this IRspectrum:
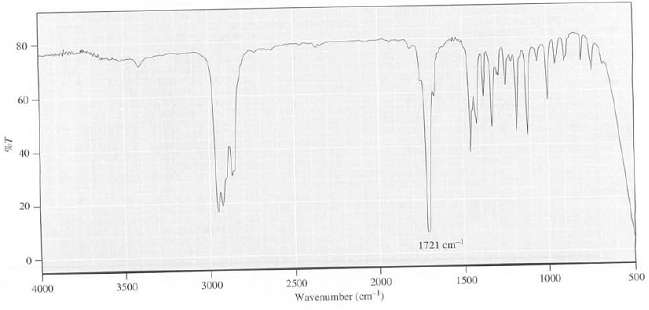
80 60 40 20 1721 cm 3500 4000 3000 2500 1500 2000 1000 S00 Wavenimber (cm)
Step by Step Solution
3.37 Rating (153 Votes )
There are 3 Steps involved in it
The intense peak at 1721 cm 1 shows the presence of a CO group The absence ... View full answer

Get step-by-step solutions from verified subject matter experts
Document Format (1 attachment)
15-C-O-S (60).docx
120 KBs Word File


