Explain which route would provide a better synthesis of theseethers: CH3 CH, CH3 CHI + CH,CO a)
Question:
Explain which route would provide a better synthesis of theseethers:
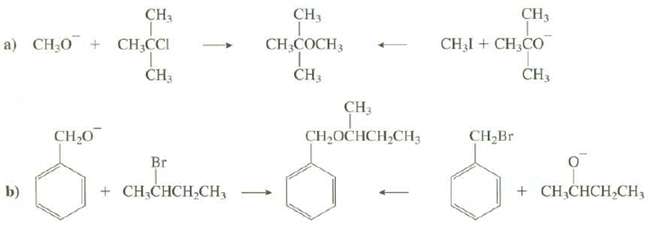
Transcribed Image Text:
CH3 CH, CH3 CHẠI + CH,CO a) CH.O + CH,CCI CH3 CH,COCH3 ČH3 CH3 CH, CH,OCHCH,CH; CH,Br CH,0 Br + CH;CHCH,CH3 b) + CH;CHCH,CH,
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 75% (8 reviews)
a The right route is better because methyl iodide cannot undergo an e...View the full answer

Answered By

Bree Normandin
Success in writing necessitates a commitment to grammatical excellence, a profound knack to pursue information, and a staunch adherence to deadlines, and the requirements of the individual publication. My background comprises writing research projects, research meta-analyses, literature reviews, white paper reports, multimedia projects, reports for peer-reviewed journals, among others. I work efficiently, with ease and deliver high-quality outputs within the stipulated deadline. I am proficient in APA, MLA, and Harvard referencing styles. I have good taste in writing and reading. I understand that this is a long standing and coupled with excellent research skills, analysis, well-articulated expressions, teamwork, availability all summed up by patience and passion. I put primacy on client satisfaction to gain loyalty, and trust for future projects. As a detail-oriented researcher with extensive experience surpassing eight years crafting high-quality custom written essays and numerous academic publications, I am confident that I could considerably exceed your expectations for the role of a freelance academic writer.
5.00+
7+ Reviews
21+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Explain which of the reaction would provide a better synthesis of3-hexanone. . . CH,CH,CH,CH,CH, 3-Hexanone CH,CH,C=CCH CH3 CH,C=CCH,CH,CH3 H,SO4 HgSO, H&SO,
-
Explain which of these reactions would provide a better synthesis of2-pentene: Br CH,OH CH,CH,CHCH,CH, + CH;0 CH,CH=CHCH,CH3 Br CH, CH CH=CHCH CH, CH,CHCH,CH,CH; + CH,0
-
Provide a retrosynthetic analysis and synthesis for each of the following compounds. Permitted starting materials are phenylmagnesium bromide, oxirane, formaldehyde, and alcohols or esters of four...
-
Evaluate the integral (4e* + 2 In (2))dx.
-
Some businesses use only approximations when estimating cost functions. What are some possible reasons for this?
-
Apple Academy is a profit-oriented education business. Apple provides remedial training for high school students who have fallen behind in their classroom studies. It charges its students $750 per...
-
Consider the vector \(u\) that consists of 32 equally spaced samples of the function \(f(t) \approx \cos (4 \pi t)\) on the interval [0,1]. That is, \(u_{1}=f(0), u_{2}=\) \(f\left(\frac{1}{32}...
-
Below are listed various costs that are found in organizations. 1. Hamburger buns in a Wendys outlet. 2. Advertising by a dental office. 3. Apples processed and canned by Del Monte. 4. Shipping...
-
You are saving up for your child's braces. You know you need $5,000 in 3 years when all his adult teeth have grown in. If your bank offers 4% interest, how much must you deposit today? Write a...
-
A teacher has given four tests to a class of five students and stored the results in the following matrix Discuss methods of matrix multiplication that the teacher can use to obtain the information...
-
Diphenhydramine can also be synthesized by heating bromo diphenyl methane with the amino alcohol shown here. Offer a reason why the oxygen, rather than the nitrogen, of this compound acts as the...
-
Suggest a synthesis of these ethers starting with an alcohol and an alkylhalide: OCH CH,CH a) CH,OCH,CH,CH,CH, b) c)
-
Shelly Mills, who has considerable experience in the home security industry, is planning to launch a firm that will sell a new line of home security alarms that she believes will be superior to...
-
Define internal combustion engine and explain how it is different from external combustion engines?
-
A thin disk of radius \(R=62.5 \mathrm{~mm}\) has uniform surface charge density \(\sigma=7.5 \mathrm{nC} / \mathrm{m}^{2}\). Calculate the potential on the axis of the disk at distances (a) 5.0 mm...
-
A particle carrying a charge of \(-5.0 \mu \mathrm{C}\) is located at the origin of a rectangular coordinate system, and a particle carrying a charge of \(12.0 \mu \mathrm{C}\) is located at \((1.0...
-
Explain the fundamental differences between Otto cycle and Diesel cycle mentioning the advantages over each other.
-
Two nonconducting spheres 1 and 2 carry the same charge, and the magnitude of the electric force exerted by each sphere on the other is \(0.10 \mathrm{~N}\) when they are \(50 \mathrm{~mm}\) apart....
-
Find all possible I/O equations for a system with state-space form where [x = Ax+ Bu y=Cx + Du
-
What does non-recourse financing mean?
-
Solve each problem. 290% of 137 miles is what?
-
Rank the following dienophiles in order of their expected reactivity in the Diels?Alder reaction. CH CH NC NC CN c=C CH c=c c=C c=C NC CN
-
1, 3-Cyclopentadiene is very reactive in DielsAlder cyclo addition reactions, but 1, 3-cyclohexadiene is less reactive and 1, 3-cycloheptadiene is nearly inert. Explain. (Molecular models are...
-
1, 3-Pentadiene is much more reactive in Diels?Alder reactions than 2, 4-pentadienal. Why might this be? H 1,3-Pentadiene 2,4-Pentadienal
-
Suppose the correlation between the stock euro returns of Siemens and the USD/EUR exchange rate is 0.2. The standard deviation of the USD/EUR is 10% and the standard deviation of Siemens's stock euro...
-
list and describe the three key client-related factors that the advisor is required to consider when developing a "suitable" investment portfolio for their client. Please cite resources used
-
Year 1 2 3 Amount ($) 2000 3000 4000 An investment made today will pays you the above cash flows at the end of each year. If your required rate of return is 5% annual interest, how much will you pay...

Study smarter with the SolutionInn App


