Explain why the reaction of the cis-isomer of this compound with potassium tert-but oxide in tert-butanol is
Question:
Explain why the reaction of the cis-isomer of this compound with potassium tert-but oxide in tert-butanol is about 500 times faster than that of thetrans-isomer.
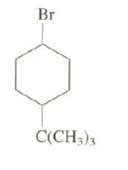
Transcribed Image Text:
Br C(CH3)3
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (14 reviews)
The most stable chair conformation of the trans isomer has b...View the full answer

Answered By

Utsab mitra
I have the expertise to deliver these subjects to college and higher-level students. The services would involve only solving assignments, homework help, and others.
I have experience in delivering these subjects for the last 6 years on a freelancing basis in different companies around the globe. I am CMA certified and CGMA UK. I have professional experience of 18 years in the industry involved in the manufacturing company and IT implementation experience of over 12 years.
I have delivered this help to students effortlessly, which is essential to give the students a good grade in their studies.
3.50+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Explain why the reaction shown in eq. 7.19 occurs much more easily than the reaction (That is, why is it necessary to protonate the alcohol before ionization can occur?) (CH) C-OH(CHCHO
-
Explain why compound A reacts faster than compound B when they undergo solvolysis in aqueous acetone. CH C-Cl CH3 CH CH
-
a. Explain why the reaction of an alkyl halide with ammonia gives a low yield of primary amine. b. Explain why a much better yield of primary amine is obtained from the reaction of an alkyl halide...
-
A major electronics manufacturer expects to generate additional revenue from its recently won government contract. The company forecasts that the revenue will be $190 million in the first year, but...
-
Refer to the 'Real life scenario describing the cost of Australian wine. Do you think Australian winemakers use job costing, process costing or a combination of these two product costing systems?...
-
Jackson et al. (1987) compared the precision of systematic and stratified sampling for estimating the average concentration of lead and copper in the soil. The 1-km2 area was divided into 100-m...
-
A jet-propelled airliner flies at an altitude of \(15,000 \mathrm{~m}\) and a velocity of \(265 \mathrm{~m} / \mathrm{s}\). What is the stagnation pressure and stagnation temperature of the air...
-
Presented below and on the next page are the financial statements of Cheaney Company. Additional data: 1. Dividends declared and paid were $25,000. 2. During the year equipment was sold for $8,500...
-
17. In the past, Marcie's father rode his bike 108 miles in 7.5 hours. Her mother rode the same distance in 8 hours. Marcie plans to ride her bike 108 miles at a steady rate of 18 mph for y hours....
-
5,000 kg/h of water will be heated from 20C to 35C by hot water at 140C. A 15C hot water temperature drop is allowed. A number of double-pipe heat exchangers, each connected in series, will be used....
-
Show the product of thisreaction: Ph Br- - + NaOEt Br EIOH Ph
-
Explain which of these compounds has a faster rate of E2elimination: CH CH3 CI 'CI
-
Vincent Corp. has 100,000 share of $100 par common stock outstanding. On June 30, Vincent Corp declared a 5% stock dividend to be issued July 30 to stockholders of record July 15. The market price of...
-
Calculate the interquartile range for each of the following sets of data: a. \(1,2,2,3,4,5,7,10\) b. \(21,8,17,7,12,19,5,12\) c. \(6,9,5,14,3,15,19,7,13,6,8,5\) d....
-
You have collected the following data: If you randomly select one of these 10 numbers, what is the probability the number (X) will be ... a. equal to 4 ? b. equal to 7 ? c. less than 5 ? d. greater...
-
You have collected the following data: If you place these five numbers in a bag and randomly select one, what is the probability the number (X) will be ... a. equal to 6 ? b. less than 11 ? c....
-
You go out to the best restaurant in town and order a steak dinner for 40. After eating half of the steak, you realize that you are quite full. Your partner wants you to finish your dinner, because...
-
For each of the following situations, calculate the population standard error of the mean \(\sigma \mathrm{X}^{-}\). a. \(\sigma=18 ; N=36\) b. \(\sigma=9.42 ; N=49\) c. \(\sigma=1.87 ; N=60\) d....
-
How to interpret the coefficient on an indicator variable in a log-linear equation.
-
Find the APR in each of the following cases: NUMBER OF TIMES COMPOUNDED Semiannually Monthly Weekly Infinite EAR APR 10.4% 8.9 11.6 15.4
-
Find the slope of each line. 4 x
-
Is a nucleus that absorbs at 6.50 more shielded or less shielded than a nucleus that absorbs at 3.20 ? Does the nucleus that absorbs at 6.50 require a stronger applied field or a weaker applied...
-
Identify the indicated sets of protons as unrelated, homotopic, enantiotopic, ordiastereotopic: (a) (b) C (c)
-
How many types of nonequivalent protons are present in each of the followingmolecules? (c) (b) CH3CH2CH20CH3 (a) H3C CH3 Naphthalene (e) (d) C=CH2 CO2CH2CH3 Ethyl acrylate Styrene
-
123 Anna purchased 100 shares of Delta stock on February 1, Year 2, for $46 per share, and 5 received a two-for-one stock split on December 31, Year 2. Anna sold all the shares of Delta stock on...
-
work i Saved QS 17-10 (Algo) Computing activity rates for activity-based costing LO P3 A company sells two types of products: standard and deluxe. It prepares the following analysis showing budgeted...
-
! Required information [The following information applies to the questions displayed below.] Sweeten Company had no jobs in progress at the beginning of the year and no beginning inventories. It...

Study smarter with the SolutionInn App


