Find the value of n?in Equation 38-32 that gives the measured dissociation energy of 741 kJ/mol for
Question:
Find the value of n?in Equation 38-32 that gives the measured dissociation energy of 741 kJ/mol for LiCl, which has the same structure as NaCl and for which r0 = 0.257 nm.
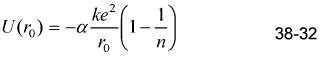
Transcribed Image Text:
ke? (1-) U(r.) = -a- 38-32
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 80% (10 reviews)
Using Equations 3832 and 38...View the full answer

Answered By

HARSH RANJAN
Taken classes at college to graduates, Also worked as an expert to a freelancer online question-solving portal for more than 8 months with an average rating greater than 4.2 out of 5.
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Fundamentals of Ethics for Scientists and Engineers
ISBN: 978-0195134889
1st Edition
Authors: Edmund G. Seebauer, Robert L. Barry
Question Posted:
Students also viewed these Modern Physics questions
-
Find the value of n in each proportion. a. 2.54cm/1in = n/12in b. 1km/0.625mi = n/200mi c. 1yd/0.96m = 140yd/n
-
Find the value of permutation. 6P2
-
Find the value of permutation. 7P2
-
In a 1994 study, 164 pregnant, HIV-positive women were randomly assigned to receive the drug AZT during pregnancy and 160 such women were randomly assigned to a control group that received a placebo....
-
Most companies use the voucher system. Due to poor profitability, several employees in the accounting office were let go. Joe Rose, who handled the verification of the vouchers in the voucher...
-
Identify several career fields that are likely to be negatively impacted by artificial intelligence applications. Can you identify any career fields that might be positively impacted?
-
Pair off in class and practice a salespersons approach to a customer. Develop an approach that lasts 3 minutes and includes a greeting.
-
Stewart Recording Studio, owned by Ron Stewart, showed the following bank reconciliation at March 31: A list of deposits made and cheques written during April, taken from the Cash Receipts Journal...
-
Required information [The following information applies to the questions displayed below.] Camille Sikorski was divorced in 2018. She currently provides a home for her 15-year-old daughter Kaly. Kaly...
-
Quest, Inc. is a calendar-year, accrual-basis C corporation engaged in manufacturing. Listed in column A are year 2 transactions from Quests financial statements and tax records. In column B, enter...
-
The distance between the Li+ and Cl ions in LiCl is 0.257 nm. Use this and the molecular mass of LiCl (42.4 g/mol) to compute the density of LiCl.
-
Suppose identical bowling balls of radius R are packed into a hexagonal close-packed structure. What fraction of the available volume of the unit cell is filled by the bowling balls?
-
Jallouk Corporation has two different bonds currently outstanding. Bond M has a face value of $20,000 and matures in 20 years. The bond makes no payments for the first six years, then pays $900 every...
-
Based on the assessment of Rent The Runway's business in January 2010, if you were Hyman or Fleiss, would you proceed with scaling (accelerated business growth) at this time? What are the...
-
Give a discussion on the following: Why is intellectual Property (IP) under attack. Nation-states, such as China and Russia, are always targeting IP within the U.S., whether it's a commercial...
-
Explain the case for integrating knowledge from OB and OA. To what extent do you feel that failing to integrate knowledge from both areas can result in an incomplete or inaccurate explanation of...
-
8. Show that a term structure model with flat yield curves and parallel yield curve shifts contains arbitrage opportunities using the following model: At time 0 the yield curve is flat at 8%. At time...
-
"The Aggregate Model of the Macro Economy," in Economics for Managers . Find two or more recent articles (post April 2020) in the Wall Street Journal or other business publications that describe...
-
During 2018, Kay Co. made the following distributions to an individual shareholder who owns 455 shares of its only class of stock. The shareholder assumed the mortgage on the property. Kay Co's...
-
Quadrilateral EFGH is a kite. Find mG. E H <105 G 50 F
-
Using a single op amp, design an amplifier with a gain of v 2 /v 1 = 3/4, input resistance of 8 k, and zero output resistance.
-
In situations where a small signal must travel over a distance, a shielded cable is used in which the signal wire is surrounded by an insulator and then enclosed by a cylindrical conductor carrying...
-
What is the advantage of placing the two insulated electric wires carrying as close together of even twisted about each other?
-
Explain why, exactly the lights may dim briefly when a refrigerator motor starts up. When an electric heater is turned on, the lights may stay dimmed as long as the heater is on. Explain the...
-
Sunland Corp. designs and builds custom harvesting equipment for logging companies across Canada. The company, which is publicly traded, has a May 31 year end. On February 18, 2024, Sunland signed a...
-
Kubin Company's relevant range of production is 18,000 to 22,000 units. When it produces and sells 20,000 units, its average costs per unit are as follows: Average Cost per Unit Fixed administrative...
-
The following balance sheet for the Los Gatos Corporation was prepared by a recently hired accountant. In reviewing the statement you notice several errors. LOS GATOS CORPORATION Balance Sheet At...

Study smarter with the SolutionInn App


