For each of the following compounds, draw an isomer that has the same functional groups. Each intersection
Question:
For each of the following compounds, draw an isomer that has the same functional groups. Each intersection of lines represents a carbon atom with the appropriate number of hydrogensattached.
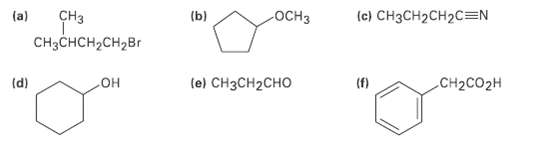
Transcribed Image Text:
(ы CHз (a) ОCHЗ (c) CH3CH2CH2C=N CH3CHCH,CH2Br le) CHзCH2CHO (d) (f) CH2CO2H он
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 66% (6 reviews)
d CH3CHCHCHCHBr CHOH CHOCH3 ...View the full answer

Answered By

Bhartendu Goyal
Professional, Experienced, and Expert tutor who will provide speedy and to-the-point solutions. I have been teaching students for 5 years now in different subjects and it's truly been one of the most rewarding experiences of my life. I have also done one-to-one tutoring with 100+ students and help them achieve great subject knowledge. I have expertise in computer subjects like C++, C, Java, and Python programming and other computer Science related fields. Many of my student's parents message me that your lessons improved their children's grades and this is the best only thing you want as a tea...
3.00+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Draw a structure for each of the following compounds in its more stable chair conformation. Explain your choice. (a) (b) CH3 CH3 CH (CH),C CHA CH, ," CH3 CH3
-
Draw enantiomers for each of the following compounds using: a. perspective formulas b. Fischer projections CH CH 1. CHjCHCH2H 2. CICH2CH2CHCH2CH3 3. CHjCHCHCHj
-
Draw a planar structure for each of the following compounds using dashed or solid wedges to show the stereochemistry of substituent groups (a) trans - 1,3 - dimethylcyclohexane (b) ( I S,2R)- I...
-
What does the following code fragment print? String \(s=\) "He11o World"; s. toUpperCase(); s. substring (6, 11); StdOut.println(s);
-
What monthly rent must she charge for each apartment to break even? Felice bought a duplex apartment at a cost of $150,000. Her mortgage payments on the property are $940 per month, $121 of which can...
-
As sole heir, Dazie receives all of Marys property (adjusted basis of $1,400,000 and fair market value of $3,820,000). Six months after Marys death in 2017, the fair market value is $3,835,000. a....
-
An engineer performed an experiment to determine the effect of $\mathrm{CO}_{2}$ pressure, $\mathrm{CO}_{2}$ temperature, peanut moisture, $\mathrm{CO}_{2}$ flow rate, and peanut particle size on the...
-
Incremental analysis involves the accumulation of information concerning a single course of action. Do you agree? Why?
-
A distinction is often made between management accounting and financial accounting. Management accounting focuses on internal reporting. It measures and reports financial and nonfinancial information...
-
You work for ConstCo, a medium-sized engineering and construction company with annual profits of about $100 million. You recently entered as a contractor into a bidding process for a large...
-
Draw a compound that: (a) Has nine primary hydrogens (b) Has only primary hydrogens
-
Give IUPAC names for the followingcompounds: H CHCH2CCH H CH CHH2CH2CH (b) (e) CHH2H2C CH (a) H-H H CH3CH2CHCH2CH,CHCH3 (e) CH H CHCH2CH2CHCH2CH CH CH-H>H3 (d)
-
Refer to the Psychological Science (Aug. 2011) study of fingerprint identification, Exercise 4.74 (p. 212). Recall that the study found that when presented with prints from the same individual, a...
-
What is the purpose of the revaluation reserve account? When should the account be debited and when will it be credited?
-
Of the 9 elements suggested by the SBA to be contained in a business plan, how many are solely focused on financial factors?
-
FAMST 192DT: Digital Theory Fall Quarter 2023 Please write brief answers (like your forum posts, approximately 200-300 words, or one large paragraph) to the following questions. Describe at least two...
-
What is the difference between an expense and an expenditure? Study your chosen CAFR to determine the expenditures and expenses noted. List these and show the expenses as a total percentage of total...
-
How do technological advancements and globalization impact the dissemination and evolution of social norms, and what challenges and opportunities do they present for social cohesion and cultural...
-
Suppose the NBPA had staged a strike at the depths of the Great Recession in 2009. What would economic conditions have done to the contract zones of the NBA and the NBPA?
-
Suppose the spot and six-month forward rates on the Norwegian krone are Kr 5.78 and Kr 5.86, respectively. The annual risk-free rate in the United States is 3.8 percent, and the annual risk-free rate...
-
You decide to cool a can of soda pop quickly in the freezer compartment of a refrigerator. When you take out the can, the soda pop is still liquid; but when you open the can, the soda pop immediately...
-
A compound with molecular formula C4H8O has a strong IR absorption at 1730 cm-1. Its mass spectrum includes key peaks at m/z 44 (the base peak) and m/z 29. Propose a structure for the compound and...
-
In the mass spectrum of 2, 6-dimethyl-4-heptanol there are prominent peaks at m/z 87, 111, and 126. Propose reasonable structures for these fragment ions.
-
In the mass spectrum of 4-methyl-2-pentanone a McLafferty rearrangement and two other major fragmentation pathways occur. Propose reasonable structures for these fragment ions and specify the m/z...
-
Start Access. Open the downloaded Access file named Exp19_Access_Ch03_ML1_Small_Business_Loans.accdb . Grader has automatically added your last name to the beginning of the filename. Note this file...
-
Commercial law in Canada list the questions should ask for this case: EMPLOYMENT Holm V. Agat Laboratories Ltd Bailey Gakhal, Brandon Longstreet, Colton Yaremko, Levi Johnson, & Nyah Foote October...
-
What are the underlying molecular processes that govern the remarkable adaptability of prokaryotes to extreme environments, such as those characterized by high temperatures, acidity, or salinity?

Study smarter with the SolutionInn App


