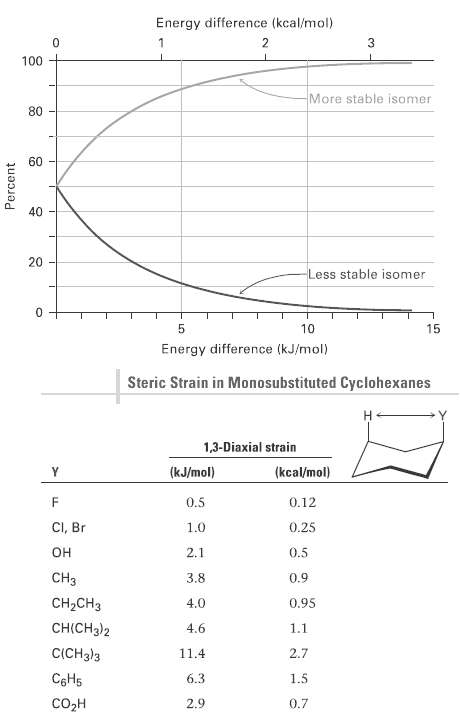
From the data in Figure and Table, estimate the percentages of molecules that have their substituents in
Question:
From the data in Figure and Table, estimate the percentages of molecules that have their substituents in an axial orientation for the following compounds:
(a) Isopropylcyclohexane
(b) Fluorocyclohexane
(c) Cyclohexane carbonitrile,C6H11CN
Transcribed Image Text:
Energy difference (kcal/mol) 2 100 More stable isomer 80 60 40 20 Less stable isomer 10 15 Energy difference (kJ/mol) Steric Strain in Monosubstituted Cyclohexanes Н 1,3-Diaxial strain (kcal/mol) (kJ/mol) 0.5 0.12 CI, Br 0.25 1.0 он 0.5 2.1 CHз 3.8 0.9 CH-CHз 4.0 0.95 CH(CH3)2 4.6 1.1 C(CH3)3 11.4 2.7 C6H5 6.3 1.5 содн 2.9 0.7 Percent
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 38% (13 reviews)
Strategy To solve this problem 1 Find the energy cost of a 13d...View the full answer

Answered By

Utsab mitra
I have the expertise to deliver these subjects to college and higher-level students. The services would involve only solving assignments, homework help, and others.
I have experience in delivering these subjects for the last 6 years on a freelancing basis in different companies around the globe. I am CMA certified and CGMA UK. I have professional experience of 18 years in the industry involved in the manufacturing company and IT implementation experience of over 12 years.
I have delivered this help to students effortlessly, which is essential to give the students a good grade in their studies.
3.50+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
From the data in Table 15.2, what can you say in general about the ratio of saturated to unsaturated acids in fats and oils?
-
The data in Figure 7.15, Table 7.19, and Table 7.20 are provided for end items A and H. Develop MRP records for items B, C, I, and E. Are there any action notices for the first week? In figure 7.15 ...
-
From the data in Table 14-6, estimate the ratio by which the yield strength of magnesium can be increased by alloying and heat treatment and compare with that of aluminum alloys.
-
With a global economy, the only way to achieve cost-effective control of greenhouse gases is to assure that every country imposes the same universal set of emissions standards. Discuss.
-
According to the financial statements for Samson Electronics, Inc., the firm has total assets valued at $310 million. It also has total liabilities of $190 million. Company records indicate that the...
-
A resistance heater operates inside a tank consuming 0.5 kW of electricity. Due to heat transfer to the ambient atmosphere at 300 K, the tank is maintained at a steady state. The surface temperature...
-
What factors must be considered in identifying the population to be sampled?
-
The following information was taken from the records of Midler Cosmetics, Inc., at December 31, 2014: Requirements 1. Using the End- of- Chapter Summary Problem (pages 637638) as an example, prepare...
-
The headlines of the National Sun Times proclaim that the current evening newscaster of channel XYZ was seen swimming with her children at the home of a well-known gentleman whose wife was currently...
-
On January 5, 2018, Alpha Inc. acquired 80% of the outstanding voting shares of Beta Inc. for $2,000,000 cash. Following are the separate balance sheets for the two companies immediately after the...
-
A Draw 1, 3, 5-trimethy1cyclohexane using a hexagon to represent the ring. How many cisTrans stereoisomers are possible?
-
A Assume that you have a variety of cyclohexanes substituted in the positions indicated. Identify the substituents as either axial or equatorial. For example, a 1, 2-cis relationship means that one...
-
Use Simpsons Rule with n = 10 to estimate the area of the surface obtained by rotating the sine curve in Exercise 7 about the x-axis.
-
What % asset allocation should one set up for oneself assuming they have just graduated? Explain why u picked what u did.
-
Do conventional financial statements satisfy the objectives of financial reporting? The financial statements that follow were adapted from those of the University of Arizona. Both the state- ment of...
-
1- Paragraph 26 of AU-C 315 requires the auditor to "identify and assess the risks of material misstatement at the ... financial statement level and levels of assertion." What are the identifiable...
-
Study the scenario and complete the question(s) that follow. Geotrack Limited You are a partner in a medium-sized audit firm, KLMP Registered Auditors, and were approached by the board of directors...
-
Use the Present Value of $1 table to determine the present value of $1 What is the total present value of the cash flows received received one year from now? Assume a 6% interest rate. Present Value...
-
Define the beta of a security.
-
At 31 December 20X9, the end of the annual reporting period, the accounts of Huron Company showed the following: a. Sales revenue for 20X9, $ 2,950,000, of which one- quarter was on credit. b....
-
Calculate the vapor pressure of water at 35.0 C using data from Tables 12.4 and 12.5. Tables 12.4 Tables 12.5 TABLE 12.4 Intermolecular Forces and Enthalpies of Vaporization at 298 Ka Liquid Dimethyl...
-
(a) Draw resonance structures for the carbocation that could be formed from (E)-2-butenyl trifluoromethanesulfonate. (b) One of the resonance structures for this carbocation should be a more...
-
What products would you expect from the reaction of 1 mol of 1,3-butadiene and each of the following reagents? (If no reaction would occur, you should indicate that as well.) (a) 1 mol of Cl2 (b) 2...
-
Provide the reagents necessary to transform 2,3-dimethyl-1,3-butadiene into each of the following compounds. (a) (b) (c) (d) Br
-
HERE IS THE SITUATION. With attendance plummeting by 50 percent in the past decade, a new, younger executive director was hired to reverse the trend. By understanding the importance of integrating...
-
Find a linear equation for the line that passes through (7,5) and (3,17)
-
Your firm has just been retained by Harley Quinn (38 years old) for a divorce and custody case. She and John Joker (42 Years Old) have been married for 10 years. She is currently residing in the...

Study smarter with the SolutionInn App


