Calculate the vapor pressure of water at 35.0 C using data from Tables 12.4 and 12.5. Tables
Question:
Calculate the vapor pressure of water at 35.0 °C using data from Tables 12.4 and 12.5.
Tables 12.4
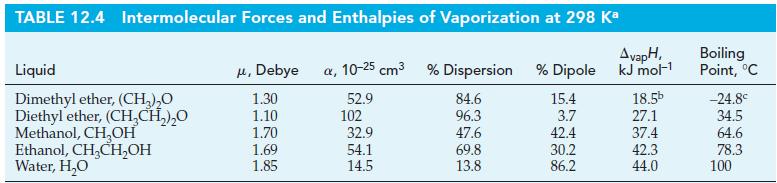
Tables 12.5
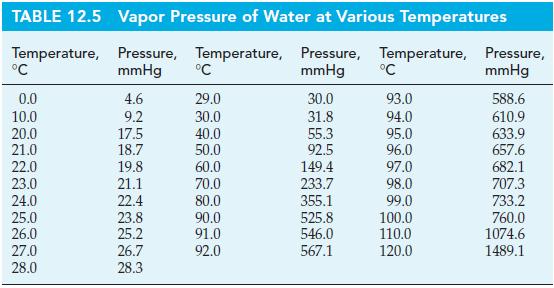
Transcribed Image Text:
TABLE 12.4 Intermolecular Forces and Enthalpies of Vaporization at 298 Ka Liquid Dimethyl ether, (CH₂)₂0 Diethyl ether, (CH₂CH₂)₂0 Methanol, CH₂OH Ethanol, CH₂CH₂OH Water, H₂O μ, Debye a, 10-25 cm³ % Dispersion % Dipole 52.9 84.6 96.3 47.6 69.8 13.8 1.30 1.10 1.70 1.69 1.85 102 32.9 54.1 14.5 15.4 3.7 42.4 30.2 86.2 AvapH, kJ mol-¹ 18.5b 27.1 37.4 42.3 44.0 Boiling Point, °C -24.8° 34.5 64.6 78.3 100
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 0% (1 review)
Analyze Starting with the ClausiusClapeyron equation we recognize that we need four pieces of data t...View the full answer

Answered By

Charles mwangi
I am a postgraduate in chemistry (Industrial chemistry with management),with writing experience for more than 3 years.I have specialized in content development,questions,term papers and assignments.Majoring in chemistry,information science,management,human resource management,accounting,business law,marketing,psychology,excl expert ,education and engineering.I have tutored in other different platforms where my DNA includes three key aspects i.e,quality papers,timely and free from any academic malpractices.I frequently engage clients in each and every step to ensure quality service delivery.This is to ensure sustainability of the tutoring aspects as well as the credibility of the platform.
4.30+
2+ Reviews
10+ Question Solved
Related Book For 

General Chemistry Principles And Modern Applications
ISBN: 9780132931281
11th Edition
Authors: Ralph Petrucci, Jeffry Madura, F. Herring, Carey Bissonnette
Question Posted:
Students also viewed these Sciences questions
-
The vapor pressure of water at various temperatures follows: (a) Draw a scatter diagram of these data. What type of relationship seems appropriate in relating y to x? (b) Fit a simple linear...
-
In the process of distillation, a mixture of two (or more) volatile liquids is first heated to convert the volatile materials to the vapor state. Then the vapor is condensed, reforming the liquid....
-
Suppose you were in charge of establishing economic policy for a small island country whose only industry is trapping. When people aren't trapping, they're enjoying leisure time with their families....
-
Suppose that a 7% semi-annual coupon bond with a time to maturity of 8 years and a par value of $100 has a price of $106,4. This bond is first callable in 6 years at a redemption price of $104,8....
-
In April, F. W. Lang Company purchased an ice cream freezer and refrigeration compressor unit from Fleet for $2,160. Although the parties agreed to a written installment contract providing for an...
-
Suppose a five-year, $1000 bond with annual coupons has a price of $990 and a yield to maturity of 6%. What is the bonds coupon rate?
-
Why are incentive travelers so important to the future of the tourism industry?
-
A monopolist faces a market demand curve given by Q = 70 P: The monopolists marginal revenue function is given by MR = 70 2Q: a. If the monopolist can produce at constant average and marginal costs...
-
Problem 16-19A (Algo) Using net present value and internal rate of return to evaluate investment opportunities LO 16-2, 16-3 Dwight Donovan, the president of Adams Enterprises, is considering two...
-
Fouke has three plants, one each in Orlando, Atlanta and Lexington each. Their capacities are 225,000 units, 175,000 units and 187,500 units respectively. Fouke needs to ship couches from these...
-
Recall the discussion of dew and frost formation. Do the surroundings absorb or lose heat when water vapor condenses to dew or frost? Is the quantity of heat per gram of H 2 O(g) condensed the same...
-
One of the following substances is a liquid at room temperature and the others are gaseous: CH 3 OH; C 3 H 8 ; N 2 ; N 2 O. Which do you think is the liquid? Explain.
-
Do terminal or instrumental values have a larger influence on your behavior at work? Explain.
-
reading the following statement and decide whether you agree or disagree with the statement: "The free market system is the best economic system since it is the most efficient and solves basic...
-
find the net presbf value of the project ? present value index? Net present value A project has estimated annual net cash flows of $11,250 for 10 years and is estimated to cost $42,500. Assume a...
-
Calculate the ICER for the new treatment, without adjusting for the health utility index. Assuming the $50K benchmark*, as a clinical decision maker or health policy advisor, would you recommend...
-
List the model assumptions for one-way ANOVA and briefly explain how to assess them.
-
Remember that a correctly labeled graph requires that you label all axes, curves, and equilibrium point values. The word "calculate" means you must show your work. Assume the market for Good Z is in...
-
The Grignard route for the synthesis of (CH3)3CCO2H from (CH3)3CBr (Example 10.5) is far superior to the nitrile route. Explain why.
-
U.S. households have become smaller over the years. The following table from the 2010 GSS contains information on the number of people currently aged 18 years or older living in a respondent's...
-
Calculating Project NPV you have been hired as a consultant for Pristine Urban-Tech Zither, Inc. (PUTZ), manufacturers of fine zithers. The market for, zithers is growing quickly. The company bought...
-
Project Evaluation Aguilera Acoustics (AAI), Inc. projects unit sales for a new seven-octave voice emulation implant as follows: Production of the implants will require $1,500,000 in net working...
-
Calculating Required Savings a proposed cost-saving device has an installed cost of $540,000. The device will be used in a five-year project but is classified as three-year MACRS property for tax...
-
Kindly Provide brief answers to the following: a) Why do we add floatation costs in the calculations of individual components costs? b) List and briefly explain the qualitative and quantitative...
-
Which of the following statements is correct. On average, the most efficient strategy to create value for existing shareholders is: To buy back shares To offer new products in a growing market To...
-
Which investment should I choose? Bond X: AA Corporate bond, Par=$1,000, Coupon rate=5% (semiannual coupons), 5 years to maturity Bond Y: AA Corporate bond, Par=$5,000, Coupon rate=5.5% (semiannual...

Study smarter with the SolutionInn App


