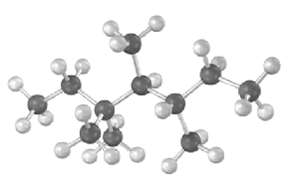
Give the IUPAC name for the following hydrocarbon, and convert the drawing into a skeletal structure.
Question:
Give the IUPAC name for the following hydrocarbon, and convert the drawing into a skeletal structure.
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 61% (13 reviews)

Answered By

Joseph Mwaura
I have been teaching college students in various subjects for 9 years now. Besides, I have been tutoring online with several tutoring companies from 2010 to date. The 9 years of experience as a tutor has enabled me to develop multiple tutoring skills and see thousands of students excel in their education and in life after school which gives me much pleasure. I have assisted students in essay writing and in doing academic research and this has helped me be well versed with the various writing styles such as APA, MLA, Chicago/ Turabian, Harvard. I am always ready to handle work at any hour and in any way as students specify. In my tutoring journey, excellence has always been my guiding standard.
4.00+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Give an IUPAC name for the following compounds: q, b.CH,CH2CHCH, c' .CH,HCHCH, CH3 CH,--C-CH3 CH3 ,
-
Give the IUPAC name for each of the following compounds: (a) CH3(CH2)25CH3 (b) (CH3)2CHCH2(CH2)14CH3 (c) (CH3CH2)3CCH(CH2CH3)2 (d) (e) (f) (g)
-
Give the IUPAC name for each of the following hydrocarbons. a. b. c. d. , , CH3 CH3 CH3CCH2CH2CH2CCH3 CH3 CH3 CH CH2CHCH2CH2CH2CH3 CH2CH2CH3 CH3 CH CHCHCH2CH2CH2 CH3 CH2CH3
-
Considerations that affect the selection of an inventory costing method do not include: (a) tax effects. (b) balance sheet effects. (c) income statement effects. (d) perpetual vs. periodic inventory...
-
How is the estate tax calculated?
-
Tests of controls. Explain what types of control tests an auditor should do in each of the following circumstances and why: 1. The auditor tests controls that contribute to the reliability of...
-
Suppose we fit the model $\mathbf{y}=\mathbf{X}_{1} \boldsymbol{\beta}_{2}+\boldsymbol{\varepsilon}$ when the true model is actually given by $\mathbf{y}=\mathbf{X}_{1}...
-
Assume that LBJ Group (LBJ), a European engineering firm, engaged in the following six transactions during the year ended December 31, Year 3. LBJ applies U.S. GAAP and reports its results in...
-
Acorn bLtd. has earnings per share of $3. It has 10 million shares outstanding and is trading at $35 per share. Acorn is considering buying a target company named Tofu Inc.. Tofu has earnings per...
-
After discarding any constants of integration, determine the appropriate value of the unknown velocities w or v which satisfy the equation of three-dimensional incompressible continuity for: (a)...
-
Name the eight 5-carbon alkyl groups you drew in Problem 3.7.
-
Make a graph of potential energy versus angle of bond rotation for propane, and assign values to the energy maxima.
-
Calculate the double integral. tan 0 = dA, R={(0, t) | 0 < 0 < T/3, 0 < t
-
What are the steps for stella assuming the application will be approved ?
-
How do legal frameworks, policy interventions, and institutional reforms contribute to shaping the direction and pace of social change, particularly in addressing systemic injustices and advancing...
-
What is Packaging & Labelling in functions of marketing? Explain.
-
Effective May 1 , the shareholders of Chiefs Corporation approved a 2 - for - 1 stock split of the companys common stock. Prior to the stock split, Chiefs shareholder s equity items were as follows:...
-
What are types of non - time series data could a finance department use to gain deeper insights via Alteryx Auto Insights?
-
Did the NFL make a mistake in allowing the Rams and Chargers to relocate to Los Angeles? Did the voters in San Diego and St. Louis make a mistake in letting them leave?
-
Listed below are several terms and phrases associated with basic assumptions, broad accounting principles, and constraints. Pair each item from List A (by letter) with the item from List B that is...
-
Without doing calculations, indicate how you would expect the lattice energies of LiCl(s), KCl(s), RbCl(s), and CsCl(s) to compare with the value of -787 kJ mol -1 determined for NaCl(s).
-
Repeat Exercise 8.26 using 1-methylcyclopentene instead of 1-butene. In Exercise 8.26 (a) HI (b) H2, Pt (c) Dilute H2SO4, warm (d) Cold concentrated H2SO4 (e) Cold concentrated H2SO4, then H2O and...
-
Write structures for the major organic products from the following reactions. Show stereoisomers where applicable. (a) (b) (c) (d) (e) H20, H.So4 dr (1) BH THF (2) NaOH, H20, Br (1) 03 (2) Meps
-
Give the structure of the products that you would expect from the reaction of 1-butyne with: (a) One molar equivalent of Br2 (b) One molar equivalent of HBr (c) Two molar equivalents of HBr (d) H2...
-
3. a. b. Solve the system by inverting the coefficient matrix and using Theorem 1.6.2 of the textbook. -x-2y-32 = 0 w+x+4y+4z = 7 w+3x+7y+9z = 4 -w-2x-4y - 6z = 6 Determine conditions on the b's, if...
-
1. If 5,000 earns Php86.00 interest in 15 months, how much is the rate of interest? 2. If the interest rate is 7.5% per year and the principal is Php2,500, determine the amount of interest for 20...
-
Given no change to the 0.8 percent chance that any women have breast cancer, and given that positive mammogram effectiveness is increased from 90% , and given that the false positive rate is...

Study smarter with the SolutionInn App


