Give the major product of each of the following reactions: a. b. c. d. e. f. g.
Question:
Give the major product of each of the following reactions:
a.
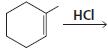
b.
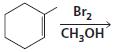
c.
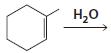
d.
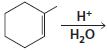
e.
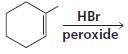
f.
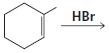
g.
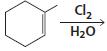
h.
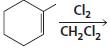
HCI CH3OH H20 Η, H+ H20 HBr peroxide HBr Cl2 H2O Cl2 CH2CI
Step by Step Answer:
a b c No rea...View the full answer



Related Video
Hydrogen peroxide can be used as a mild antiseptic to curb superficial skin infections such as athlete’s foot, but only in diluted quantities. To combat stinky feet, try soaking your feet in a solution of 1 part 3% hydrogen peroxide and 3 parts warm water for 15-20 minutes, then drying them thoroughly. This will kill odor-causing bacteria and soften your feet. To treat athlete\'s foot, you can use a similar solution, but only in diluted quantities, and soak your feet for 30 minutes. Hydrogen peroxide can also be used to keep vegetables fresh by adding 1/4 cup to a bowl of cold water, soaking the vegetables for 20-30 minutes, then draining, drying, and refrigerating them. Alternatively, you can spray vegetables with a solution of 3% hydrogen peroxide and let them stand for a few minutes before rinsing and drying. To keep leftover salad fresh, spray it with a solution of 1/2 cup water and 1 Tbsp. 3% hydrogen peroxide, drain, cover, and refrigerate.
Students also viewed these Organic Chemistry questions
-
Give the major product of each of the following reactions: a. b. c. d. e. f. HBr HC CCH3 peroxide excess Br 3 CH2Cl2 excesS CHC CCH HBr excess
-
Give the major product of each of the following reactions a. b. c. d. e. f. g. h. i. CCH3 + HNO3 S NO2 CHs CH CHCH NCH CH 3 CH3 HO CH3NCH3 + PC15- NC 1. H202 2. CH3 1. HO 3. Ht CH3 + CH:CH2MgBr 2.4...
-
Give the expected major product of each of the following reactions. PCC is the abbreviation for pyridinium chlorochromate (Section 8-6). (a) (b) (c) (d) (e) CH CH CH,OH NeCrO HSO, HO PCC, CH,CI (CH3)...
-
Formulate a plausible mechanism for the following reaction. The product is a precursor of mediquox (shown in the margin), an agent used to treat respiratory infections in chickens (no, we are not...
-
Highcountry.com is an Internet retailer of sporting good products. Customers order sporting goods from the company, using an online catalog. The company processes these orders and delivers the...
-
What five questions typically are used in fact-finding? What other question does the Zachman Framework include? Is the additional question important?
-
Visit www.pearsonglobaleditions.com/malhotra to read the video case and view the accompanying video. Marriott: Marketing Research Leads to Expanded Offerings highlights Marriotts success in using...
-
Kelton Company sells women's clothing. Kelton's strategy is to offer a wide selection of clothes and excellent customer service and to charge a premium price. Kelton presents the following data for...
-
Two speeding lead bullets, one of mass 13.0 g moving to the right at 320 m/s and one of mass 7.95 g moving to the left at 385 m/s, collide head-on, and all the material sticks together. Both bullets...
-
Suppose Good-to-Go (Problem 4-22) is considering the possible introduction of two new products to its line of suitcases: the Compact model (for teenagers) and the Kiddo model (for children). Market...
-
For each pair of bonds, which has the greater strength? Briefly explain why. a. b. c. d. CH,-Cl or CH,-Br CH,CH2CH2 or CH,CHCH3 CH, CH or CH, CH,CH, I-Br or BrBr
-
Using any alkene and any other reagents, how would you prepare the following compounds? a. b. c. d. e. f. CH3CH CH2CHCH CI CH2CHCH3 CH3CH2CHCHCH2CH3 Br CH CH2CHCHCH2CH3 Br Cl
-
Haley Jones and Olivia Dunne have used a combination of expert and referent power to earn big money through NIL deals. What does this case suggest about followership (and about how leaders might...
-
If a universal life insurance policy uses "adjustable monthly mortality deduction rates," what does that usually mean? Question 4 options: The rate charged for the insurance element of the contract...
-
Steed Company prepared a promissory note with a face value of $50,000, payable after 50 days, at a rate of 19%. Calculate the interest amount of the promissory note. Assume 365 days in a year. (Round...
-
you retire with a large amount of savings in aretirement account. You decide to withdraw 3.5% to 4% of your savings the first year and increase the rate by inflation each year. What is the basis of...
-
Question 20 (1 point) Common types of taxes one pays in Canada are: Question 20 options: GST, HST & PST Property Tax Income Taxes All of the above
-
Where would you place Ferrari in regards to using an EBITDA multiple for valuation purposes? What EBITDA multiple would you ascribe to Ferrari?
-
In Problem find the domain of the function and all x or y intercepts. 3 f(x) x? - 1 ||
-
Wilsons Auto Repair ended 2011 with Accounts Receivable of $85,000 and a credit balance in Allowance for Uncollectible Accounts balance of $11,000. During 2012, Wilsons Auto Repair had the following...
-
Trans-1-Phenyl-1, 3-butadiene has max = 280 ( = 27,000) calculate the concentration of a solution that has A = 0.643 at 280nm in a 1 cm cell.
-
Nitro methane max = 275nm ( = 1.5) what kind of transition is responsible for this absorption?
-
3-Buten-2-one has max =213nm ( = 7080) and max = 320nm ( = 21) what kind of transition is responsible for each of these absorptions?
-
Watch the recorded interview of Ms . Veronica Trammell, Executive Director of Learning Technology at KSU. The interview focused on service management and service operation. Share your thoughts in...
-
develop a detailed event plan following the assigned event type and theme with the following information: Event objectives based on the assigned event type and theme Two sets of varying food &...
-
Provide a detailed simulation performance results for Walmart inc. based on 5 year performance review.

Study smarter with the SolutionInn App


