Give two Friedel-Crafts acylation reactions that could be used to prepare 4-methoxybenzophenone. Which reaction would be faster
Question:
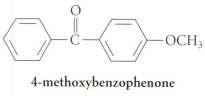
Give two Friedel-Crafts acylation reactions that could be used to prepare 4-methoxybenzophenone. Which reaction would be faster or occur under milder conditions? Explain.
Transcribed Image Text:
OCH3 4-methoxybenzophenone
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 84% (13 reviews)
The two possibilities are the acylation of anisole by benzo...View the full answer

Answered By

OTIENO OBADO
I have a vast experience in teaching, mentoring and tutoring. I handle student concerns diligently and my academic background is undeniably aesthetic
4.30+
3+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Show two different Friedel-Crafts acylation reactions that can be used to prepare the following compound. CH3 CH CH3
-
Show reactions that could be used to convert 1-butanol to thesecompounds: b) a) CI NH2 d) c) SCH CH " f) e) OCH h) g) CN j) i) OCCH-CH3
-
Show reactions that could be used to convert the epoxide to these compounds. More than one step may benecessary. SCH3 OH b) CH,O- a) Br
-
A nutritionist at the Food and Drug Administration is studying the effects of cereal marketing on family meal choices. In particular, she would like to understand how cereal manufacturers market...
-
East Company leased a new machine from North Company on May 1, 2010 under a lease with the following information: Lease Term 10 years Annual rental payable at the beginning of each lease year $40,000...
-
What are the main reasons of using a VPN?
-
National Life Expectancies. We continue the analysis begun in Exercises 1.7and 2.22 Now fit a regression model on LIFEEXP using three explananatural logarithmic transform of PRIVATEHEALTH). a....
-
On March 1, 2014, Quinto Mining Inc. issued a $600,000, 8%, three-year bond. Interest is payable semi-annually beginning September 1, 2014. Required Part 1 a. Calculate the bond issue price assuming...
-
2-1. GASB Concepts Statement No. 3 discusses various methods of communicating financial accounting information to users. One method is using required supple- mentary information (RSI). What is RSI,...
-
Are middle-class families more likely than working- class families to maintain contact with kin? Write a paragraph summarizing the results of these tests. a. A sample of middle-class families...
-
Rank the following compounds in order of increasing reactivity in bromination. In each case, indicate whether the principal monobromination products will be the ortho and para isomers or the meta...
-
Give the structures of all the hydrocarbons C10H10 that would undergo catalytic hydrogenation to give p-di-ethylbenzene.
-
The production budget of Rooney Company calls for 80,000 units to be produced. If it takes 30 minutes to make one unit and the direct labor rate is $16 per hour, what is the total budgeted direct...
-
In 2020, oil and gas prices plummeted, which left major oil and gas companies, such as Shell (a British multinational company listed on the London Stock Exchange) and Exxon Mobil (an American...
-
Describe how the responsibilities of a manager might differ between a cost center, a profit center, and an investment center. How do the reports differ for each?
-
Describe how the Recording Industry responded when the long play 33 1/3 (LP) format and the 45 rpm record formats were introduced, as opposed to the 78 rpm records with each song as a separate...
-
a. Performed $27,000 of services on account. b. Collected $19,100 cash on accounts receivable. c. Paid $5,200 cash in advance for an Insurance policy. d. Paid $810 on accounts payable. e. Recorded...
-
Caroline Corp purchased the assets of Hayden Inc. for $7 million cash. The fair value of Hayden Inc's assets was 3.4 million and the purchased company had total liabilities of $350,000. How much...
-
The following financial ratios and information relate to Starbucks Corporation for the period 2003 to 2012. Required a. Represent the 2012 horizontal analysis of the asset section of the balance...
-
What services are provided by the provincial and territorial governments?
-
Give the products expected when D-ribose (or other compound indicated) reacts with each of the following reagents. (a) dilute HNO3 (b) -CN, HO (c) product of part (b) + H/Pd/BaSO4 + HO+/HO (d) CHOH,...
-
Give the product(s) expected when d-mannose (or other compound indicated) reacts with each of the following reagents. (Assume that cyclic mannose derivatives are pyranoses.) (a) Ag(NH3) (b) dilute...
-
Consider the structure of cellobiose, a disaccharide obtained from the hydrolysis of the polysaccharide cellulose. Into what monosaccharide(s) is cellobiose hydrolyzed by aqueous HCl? HO CHOH -...
-
Compare life cover end funeral cover stating the advantages and disadvantages of each.
-
What are the change management issues in C 3 solutions?
-
What are 2 key events discussed in the documentary kanyi?

Study smarter with the SolutionInn App


