Grignard reagents undergo a general and very useful reaction with ketones. Methyl magnesium bromide, for example, reacts
Question:
Grignard reagents undergo a general and very useful reaction with ketones. Methyl magnesium bromide, for example, reacts with Cyclohexanone to yield a product with the formula C7H14O. What is the structure of this product if it has an IR absorption at 3400 cm?1?
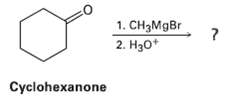
Transcribed Image Text:
1. CH3MgBr 2. H30* Cyclohexanone
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 71% (14 reviews)
CH3 OH 1 CH3MgB...View the full answer

Answered By

Amir Farooq
I have done BSC from s.p college srinagar. After that i did my MBBS from achariya shri chander college of medical science sidhra jammu.currently i am an intern doctor at skims srinagar. I love to teach students across the globe. I was teaching students of 12 th class at my room from the last 5 years.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
What is the structure of ATCase?
-
What is the structure of the global beer industry?
-
What is the structure of the beer industry?
-
Night By Elie Wiesel The Holocaust - Why did the members of Sighets Jewish community refuse to believe their horrible situation? (Moshe the Beadle and Madame Schachter portending the horrors that...
-
How a firm behaves toward existing competitors is a major determinant of whether it will face entry by new competitors. Explain.
-
A laser beam shines along the surface of a block of transparent material (see Fig. E33.8.). Half of the beam goes straight to a detector, while the other half travels through the block and then hits...
-
Who is often recognized as being the first hospital administrator and what major contributions did she make to nursing and the improvement of patient care?
-
Prepare budgetary entries, using general ledger accounts only, for each of the following unrelated situations: a. Anticipated revenues are $10 million; anticipated expenditures and encumbrances are...
-
Direct materials Average Cost per Unit $ 7.80 Direct labor $ 4.80 Variable manufacturing overhead $ 2.30 .Fixed manufacturing overhead $ 5.80 Fixed selling expense $ 4.30 Fixed administrative expense...
-
A drag strut splice between 2x4 No.2 DF-L top plate and a 4x6 No.1 DF-L header needs to resist tension force (seismic) equal to the capacity of the 2x4 in tension. 1. Determine the maximum tension...
-
Methyl-2-pentanone and 3-methylpentanal are isomers. Explain how you could tell them apart, both by mass spectrometry and by infraredspectroscopy. H. 4-Methyl-2-pentanone 3-Methylpentanal
-
Ketones undergo a reduction when treated with sodium borohydride, NaBH 4 . What is the structure of the compound produced by reaction of 2-butanone with NaBH4 if it has an IR absorption at 3400 cm ?1...
-
1. When a Hong Kong fi rm makes an agreement with the Walt Disney Company to use the Disney logo and legally make jewelry in the shape of Disney cartoon characters, Disney is engaging in a form of...
-
Valuation of a constant growth stock A stock is expected to pay a dividend of $2.50 the end of the year (that is, D1 = $2.50), and it should continue to grow at a constant rate of 7% a year. If its...
-
Do you know someone who acts a lot like Eeyore from Winnie the Pooh? Everything is ho hum, depressing or too hard. Maybe you work with a super happy person who seems to have one setting...shiny! Our...
-
explain column exchange chromoyagraphy how amino acid content is identified, with drawing and simple step Explanation
-
Valuation of a declining growth stock Martell Mining Company's ore reserves are being depleted, so its sales are falling. Also, because its pit is getting deeper each year, its costs are rising. As a...
-
How do advancements in genomic sequencing technologies contribute to the identification of target genes for therapeutic intervention, and how does this inform the development of personalized gene...
-
A researcher wants to know if there is a difference in the average number of hours a person sleeps based on four different types of natural sleep supplements (melatonin, herbal tea, tart cherry...
-
(8%) Problem 6: A student attaches a f= 3.5 kHz oscillator to one end of a metal rail of length L = 25 m. The student turns on the oscillator and uses a piezoelectric gauge at the other end to...
-
Potassium perchlorate (KClO 4 ) has a lattice energy of -599 kJ/mol and a heat of hydration of -548 kJ/mol. Find the heat of solution for potassium perchlorate and determine the temperature change...
-
When cis-2-methylcyclohexanol reacts with the Lucas reagent, the major product is 1-chloro- 1-methylcyclohexane. Propose a mechanism to explain the formation of this product?
-
Write balanced equations for the three preceding reactions?
-
Suggest how you would convert trans-4-methylcyclohexanol to (a) Trans-1-chloro-4-methylcyclohexane? (b) Cis-1-chloro-4-methlcycloexane?
-
There are 42 locations throughout the northeast region with deposits totaling $3.2bil. If you were Kiamarie's vice president, how would you expect her to choose between Gagne's Nine Events of...
-
Why is succession planning essential in an organization? 2. What is the need for succession planning? 3. What do you think are the challenges to succession planning? 4. How can employees contribute...
-
Two activities/obligations/responsibilities that might be added to an updated version of the job description for an HR professional: How would you go about monitoring changes in laws and regulatory...

Study smarter with the SolutionInn App


