Ketones undergo a reduction when treated with sodium borohydride, NaBH 4 . What is the structure of
Question:
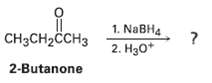
Ketones undergo a reduction when treated with sodium borohydride, NaBH4. What is the structure of the compound produced by reaction of 2-butanone with NaBH4 if it has an IR absorption at 3400 cm?1 arid M+ = 74 in the mass spectrum?
Transcribed Image Text:
1. NABH4 2. Hзо* |CH3CH2CCHЗ 2-Butanone
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 53% (13 reviews)
CH3CH2CCH3 1 NaBH4 2 H3O OH ...View the full answer

Answered By

David Ngaruiya
i am a smart worker who concentrates on the content according to my clients' specifications and requirements.
4.50+
7+ Reviews
19+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
What is the structure of ATCase?
-
What is the structure of the global beer industry?
-
What is the structure of the beer industry?
-
What do you think people would say about Corrie from the few quotes we have from her book? What was her personality like? Do you think she handled her incarceration differently than Elie Wiesel?...
-
Industries with high barriers to entry often have high barriers to exit. Explain.
-
As shown in Fig. E33.11, a layer of water covers a slab of material Xin a beaker. A ray of light traveling upward follows the path indicated. Using the information on the figure, find (a) The index...
-
Although everyone makes mistakes, describe how history repeats itself when there are patterns of progress and regression in the provision of patient care. In forming your answer, consider how...
-
Glen Pool Club, Inc., has a $ 150,000 mortgage liability. The mortgage is payable in monthly installments of $ 1,543, which include interest computed at an annual rate of 12 percent (1 percent...
-
P3.1 (LO 1, 2, 3) (Multiple-Step Statement, Retained Earnings Statement) The following information is related to Dickinson Company for 2025. Retained earnings balance, January 1, 2025 Sales revenue...
-
Feng is the owner of a small business. When Feng has worked hard (a1) during the year, net income before manager compensation has been $ 1,600 60% of the time and $ 400 40% of the time. More...
-
Grignard reagents undergo a general and very useful reaction with ketones. Methyl magnesium bromide, for example, reacts with Cyclohexanone to yield a product with the formula C 7 H 14 O. What is the...
-
Nitriles, RC N, undergo a hydrolysis reaction when heated with aqueous acid. What is the structure of the compound produced by hydrolysis of propane nitrile, CH3CH2C N, if it has IR absorptions at...
-
Pea aphids, Acyrthosiphon pisum, can be red or green. Weirdly, red aphids make carotenoids (red pigments) with genes that jumped from a fungus into the aphid genome some time during recent...
-
What ethical considerations arise from the ability to manipulate the human genome through gene therapy, particularly concerning issues of informed consent, genetic discrimination, and equitable...
-
Find the solution of the initial value problem y" + 4y +5y= 0, Y 0, = 4. y(t) = = How does the solution behave as t o? Choose one
-
What are the primary mechanisms underlying gene therapy, and how do they differ from traditional pharmacological interventions?
-
what ways do preclinical models, including animal studies and in vitro assays, contribute to the validation and optimization of gene therapy approaches before transitioning to human clinical trials?
-
Calculate the materials cost per product using the activity-based costing method given this information: Materials cost: $440 Number of products made: 1,200
-
In problem \#5, we used the given sample data to estimate whether there is a difference in the mean number of hours slept based on the type of natural supplement used. Using Fisher's LSD, describe...
-
a. What is the cost of borrowing if Amarjit borrows $28 500 and repays it over a four-year period? b. How many shares of each stock would he get if he used the $28 500 and invested equally in all...
-
The solubility of carbon tetrachloride (CCl 4 ) in water at 25 C is 1.2 g/L. The solubility of chloroform (CHCl 3 ) at the same temperature is 10.1 g/L. Why is chloroform almost ten times more...
-
Predict the products of the reactions of the following compounds with chromic acid and also with PCC. (a) Cyclohexanol (b) 1-methylcyclohexanol (c) Cyclopentylmethanol (d) Cyclohexanone (e)...
-
Two products are observed in the following reaction. (a) Suggest a mechanism to explain how these two products are formed. (b) Your mechanism for part (a) should be different from the usual mechanism...
-
Give the structures of the products you would expect when each alcohol reacts with (1) HCI, ZnCI2; HBr; (3) PBr3: (4) P/I2; (5) SOCI2- (a) butan-1-ol (b) 2-methylbutan-2-ol (c) 2,2-dimethylbutan-1-ol...
-
given l o g x 5 = l o g 5 x solve for x
-
Solve the formula 8 x 3 y = - 1 9 for y .
-
Q6 Find the total amount for of the current balance for each underwriter and find out top 10 richest underwriter Q7 Create a pivot table for the entire data and find out the total current balance for...

Study smarter with the SolutionInn App


