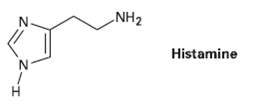
Histamine, whose release in the body triggers nasal secretions and constricted airways, has three nitrogen atoms. List
Question:
Histamine, whose release in the body triggers nasal secretions and constricted airways, has three nitrogen atoms. List them in order of increasing basicity, and explain yourordering.
Transcribed Image Text:
NH2 Histamine H.
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 53% (15 reviews)
b NH N bra CN Histamine 1 H The a nitrogen is mo...View the full answer

Answered By

Shikhar Srivastava
I have interest in science as it enables us to understand the process happening in our universe. I am a subject matter expert at Chegg solving advance physics problem with CF score more than 80% on average. I like to help student by solving problems. This helps me as well to build up my concepts regarding the subject. I have learnt from my experience that, In physics to build up command on subject one need to understand the theory, keep revising notes in free time and at last one more important thing is that one need to keep solving numerical problem related to the topic.
Taking career in core sciences is a long journey and one need to be calm minded with his or her profession.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Explain the basicity order of the following three amines: p-nitroaniline (A), rz-nitroaniline (A), and aniline (C). The structures and pKa data are shown in Table 23'l'
-
The nitrogen atoms in N2 participate in multiple bonding, whereas those in hydrazine, N2H4, do not. (a) Draw Lewis structures for both molecules. (b) What is the hybridization of the nitrogen atoms...
-
List the three states of matter in order of] (a) Increasing molecular disorder and (b) Increasing intermolecular attractions. (c) Which state of matter is most easily compressed?
-
Doug Brackett wants to have enough mechanics on hand to take care of his customer requests, but he does not want to be paying mechanics to sit around doing nothing. Doug needs to know a reasonable...
-
Based on the description in this case, how well would you say Susan Durbin appreciates the scope of human resource management? What, if any, additional skills of an HR professional would you...
-
Compute the atomic packing factor for the rock salt crystal structure in which rC/rA = 0.414.
-
Define the following terms: (i) an environmental management system; (ii) an internal environmental audit; (iii) an external verification (or assurance) engagement.
-
Be Sands purchased the business Regional Supply Co. for $285,000 cash and assumed all liabilities at the date of purchase. Regional's books showed assets of $280,000, liabilities of $40,000, and...
-
Develop a strategic business plan. The CEO of the real estate agent has given new directives regarding the increase of average profit annually. You, as a manager, have been given a job to analyse...
-
Diatomic nitrogen and oxygen molecules can stick to the surface of a container because of van der Waals forces. Assume that these molecules are stuck to the surface with binding energy...
-
Although pyrrole is a much weaker base than most other amines, it is a much stronger acid (pK a 15 for the pyrrole versus 35 for diethyl amine). The NH proton is readily abstracted by base to yield...
-
Oxazole is a live-membered aromatic heterocycle. Would you expect oxazole to be more basic or less basic than pyrrole?Explain. Oxazole N:
-
Which capital structure theories does the empirical evidence seem to support?
-
Find the curvature of r(t) at the point (1, 1, 1). r(t) = (t. 4.436 k =
-
1) Write a function named "Countstr" that operates over a list which includes a series of strings. The function takes a string and outputs the number of occurrences of that string in the given list....
-
A company has four photocopy machines A, B, C and D. The probability that a given machine will break down on a particular day is P(A) = 950 P(B) = 15 P(C) = 18/100 P(D) = 11/100 Assuming...
-
Required: 1. Prepare closing entries. 2. Prepare post-closing trial balance. The following is the unadjusted trial balance of ABC Company as the end of its second year of operations. The company...
-
The last investment option for BIC is a run-down building on 1 acre of land called Crystal Estates. They expect to rent the building as a storage facility, which will yield an NOI of $105,000 next...
-
What words comprise the acronym DCF? Describe/define what it means in 10 words or less.
-
Explain what is meant by vicarious liability and when it is available?
-
Classify each acid as strong or weak. If the acid is weak, write an expression for the acid ionization constant (K a ). a. HNO3 b. HCI c. HBr d. HSO3
-
Using structural formulas, write equations for each of the following combustion reactions For information: (see Reaction Summary 1.a ): a. The complete combustion of propane b. The complete...
-
Write the formula for each of the following compounds: a. Isobutyl chloride b. isopropyl bromide c. 2-chlorobutane d. tert-butyl iodide e. Propyl fluoride f. general formula for an alkyl bromide
-
Using structural formulas, write equations for the following halogenation reactions (see Reaction Summary 1.b, p. 63), and name each organic product: a. The monochlorination of propane b. The...
-
1-Define electric fields and how it helps us understand electricity. 2-Electric fields are represented as a physical effect of a configuration of charges that is created by the attraction of electric...
-
Why thin film on a glass surface can remove completely reflections at certain light wavelength? Magnesium fluoride (n = 1.38) is used as coating on nonreflecting glass. How thin coating is enough to...
-
How much work ( in joules) is done in lifting a 45.57 newton box from the floor to a table that is 0.73 meters above the floor? Round your final answer to two decimal places. A ball is thrown...

Study smarter with the SolutionInn App


