Oxazole is a live-membered aromatic heterocycle. Would you expect oxazole to be more basic or less basic
Question:
Oxazole is a live-membered aromatic heterocycle. Would you expect oxazole to be more basic or less basic than pyrrole?Explain.
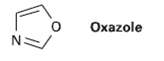
Transcribed Image Text:
Oxazole N:
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 53% (13 reviews)
B sze H Oxazole Oxazole is an aromatic 6a electron heterocycle Two oxygen electrons and one nitrogen electron ...View the full answer

Answered By

OTIENO OBADO
I have a vast experience in teaching, mentoring and tutoring. I handle student concerns diligently and my academic background is undeniably aesthetic
4.30+
3+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Would you expect heteroscedasticity to be present in the following regressions? Sample Net worth Log of net worth (a) Corporate profits Fortune 500 (b) Log of corporate (c) Dow Jones industria (o)...
-
Would you expect lysozyme to hydrolyze cellulose? Why or why not?
-
Would you expect insulin to increase or decrease the activity of the enzyme ATP-citrate lyase?
-
Kellogg Company is expected to pay $2.00 in annual dividends to its common shareholders in the future. Our best estimate of the expected cost of equity capital is 5.0% and the expected growth rate in...
-
How do talent management evidence-based HR support Mundy's efforts to offer solutions?
-
There is a road between the suburbs and downtown. The road becomes congested at rush hour. As long as fewer than 100 people use the road at rush hour, the trip takes 30 minutes. When the 101st person...
-
Consider the time to recharge the flash. The probability that a camera passes the test is 0.8, and the cameras perform independently. What is the probability that the third failure is obtained in...
-
Continue with the facts of Problem 57. What are the Federal income tax withholding requirements with respect to Martinho's sale? Who pays the withheld amount to the U.S. Treasury?
-
Let f(x)=x for rational x and f(x) = 0 for irrational x. (a) Calculate the upper and lower Darboux integrals for f on the interval [0, b]. (b) Is f integrable on [0,b]?
-
Your task is to build a JEE web application that uses Enterprise JavaBeans (EJB) and Java Persistence API (JPA) to manage the persistence of Java objects to a relational database. The application...
-
Histamine, whose release in the body triggers nasal secretions and constricted airways, has three nitrogen atoms. List them in order of increasing basicity, and explain yourordering. NH2 Histamine H.
-
Protonation of an amide using strong acid occurs on oxygen rather than on nitrogen. Suggest a reason for this behavior, taking resonance intoaccount. :0: H2SO4 NH2 NH2 R.
-
In June 2012, Karen transferred property with a $75,000 FMV and a $20,000 adjusted basis to Hal, her husband. Hal dies in March 2013; the property has appreciated to $85,000 in value by then. His...
-
Develop a marketing brochure or webpage stating all the important information a parent might need to consider your program for their child. . Including philosophy, name, address, phone, license,...
-
What term refers to the elevation of subjects of a painting by portraying them in poses and settings normally associated with positions of high status and power?
-
Simplify. Z -2 -2 4z
-
Identify key internal and external stakeholders, groups, and organizations involved in enforcing and maintaining operational sustainability regarding each aspect of the TBL.
-
An IT officer at the law firm described the system as being a type of Expert Systems technology that uses heuristics to match client cases with the thousands of previous cases in the database....
-
Determine the missing elements in the accounting equation. Treat each case separately. A LOER - EP/L Case Total Assets Total Owner's Liabilities Equity Total Total Revenues Expenses Net Profit OE at...
-
Explain how the graph of each function can be obtained from the graph of y = 1/x or y = 1/x 2 . Then graph f and give the (a) Domain (b) Range. Determine the largest open intervals of the domain over...
-
Both H 2 O and H 2 PO 4 are amphoteric. Write an equation to show how each substance can act as an acid and another equation to show how each can act as a base.
-
From the dichlorination of propane, four isomeric products with the formula C3H6Cl2 were isolated and designated A, B, C, and D. Each was separated and further chlorinated to give one or more...
-
Write all of the steps in the free-radical chain mechanism for the monochlorination of ethane. What trace by-products would you expect to be formed as a consequence of the chain terminating steps?...
-
Using methanol (CH3OH) as a fuel source, write a balanced reaction for the formation of H2. Do the same for C10H22 (a component of diesel fuel). For an H2 fuel cell, why would it be better to use...
-
Simplify. 2 x-4)2x +0x - 26x - 24
-
On January 1 , 2 0 X 2 P pays $ 1 0 0 million to acquire 5 0 , 0 0 0 of the common voting shares of S , which gives P a 2 5 % ownership interest in S . The January 1 , 2 0 X 2 book value of S ' s...
-
Shaw Company has the following account balances: Receivables $ 1 0 0 , 0 0 0 Inventory 1 5 0 , 0 0 0 Land 1 0 0 , 0 0 0 Building net 2 5 0 , 0 0 0 Liabilities 1 0 0 , 0 0 0 Common stock 1 0 0 , 0 0 0...

Study smarter with the SolutionInn App


