Horizontal analysis (trend analysis) percentages for Algy Company?s sales, cost of goods sold, and expenses are shown
Question:
Horizontal analysis (trend analysis) percentages for Algy Company?s sales, cost of goods sold, and expenses are shown below.
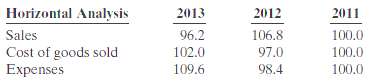
Did Algy?s net income increase, decrease, or remain unchanged over the 3-year period?
Horizontal Analysis 2013 96.2 102.0 109.6 2012 2011 100.0 100.0 Sales Cost of goods sold Expenses 106.8 97.0 98.4 100.0
Step by Step Answer:
Comparing the percentages presented results in the following conclusions T...View the full answer



Accounting Principles
ISBN: 978-0470534793
10th Edition
Authors: Jerry J. Weygandt, Paul D. Kimmel, Donald E. Kieso
Related Video
The Dupont analysis is an expanded return on equity formula, calculated by multiplying the net profit margin by the asset turnover by the equity multiplier. The DuPont analysis is also known as the DuPont identity or DuPont model.This Video will guide on how to calculate return on Equity and estimate profitability of shareholders using DuPont Analysis.
Students also viewed these Managerial Accounting questions
-
Suppose you are buying a car of $26000. The dealer offers that you pay a down payment of $4000 now. Then you will pay the balance with equal monthly payments for 42 months with 6% APR compounded...
-
The Robinson Company had a cost of goods sold of $1,000,000 in 2014 and $1,200,000 in 2015. a. Calculate the inventory turnover for each year. Comment on your findings. b. What would have been the...
-
Cubitene is a diterpene present in the defense secretion of a species of African termite. What unusual feature characterizes the joining of isoprene units in cubitene?
-
Use regression analysis to fit a quadratic trend model to the data set. a. What is the estimated regression function? b. Compare the adjusted-R2 value for this model to that of the linear trend...
-
(See Exercise 21.21.) (a) Calculate the electric potential energy of the adeninethymine bond, using the same combinations of molecules (O-H-N and N-H-N) as in Exercise 21.21. (b) Compare this energy...
-
A well-designed management information system using EDP equipment will include methods of assuring that the data are appropriate to the situation and are accurate. Required: a. Describe procedures...
-
Data for Andrews Inc. are presented in E23-13. Prepare a statement of cash flows using the indirect method.
-
Suppose that Sony sold $1,040,000, 5-year, 7% bonds on January 1, 2025 for $1,008,000. The company uses straight-line amortization. Complete the following bond amortization schedule for 2025 and...
-
Hanks Hotel opened for business on May 1, 2020. Its trial balance before adjustment on May 31 is as follows. In addition to those accounts listed on the trial balance, the chart of accounts for Hanks...
-
If Vansittart Company had net income of $585,000 in 2013 and it experienced a 30% increase in net income over 2012, what was its 2012 net income?
-
Selected condensed data taken from a recent balance sheet of Evangelista Inc. are as follows. EVANGELISTA INC. Balance Sheet (partial) Cash..............$ 8,041,000 Short-term...
-
A soccer ball is kicked from ground level with (x, y)-coordinates (85, 20) on the soccer field shown in Figure 11 and with an initial velocity v 0 = 10i 5j + 25k ft/s. Assume an acceleration of 32...
-
Given the following source code: for(int j = 1; j
-
What are some of the co-cultures that you strongly identify with? How long have you been a member of this co-culture? What are some of the elements that help define this as a co-culture?
-
What is Laissez-Faire Leadership and provide an example ?
-
What is the output of the following program? Assume d=6.4
-
Who is a great example of a Transactional Leadership and Laissez-Faire Leadership in the 20 century?
-
What are four things that you need to consider when choosing information, data, and/or demonstrations for a presentation?
-
Create a data model for one of the processes in the end-of-chapter Exercises for Chapter 4. Explain how you would balance the data model and process model.
-
For the reaction K eq = 3.59 at 900C. After the reaction has run for 10 min at 900 C, the concentrations are [CH 4 ] = 1.15 M; [H 2 S] = 1.20 M; [CS 2 ] = 1.51 M; [H 2 ] = 1.08 M. Is this reaction at...
-
What are the strengths of the annual rate of return approach? What are its weaknesses?
-
What are the strengths of the annual rate of return approach? What are its weaknesses?
-
Your classmate, Mike Dawson, is confused about the factors that are included in the annual rate of return technique. What is the formula for this technique?
-
What are some design considerations for a document database versus a relational database Explain CRUD in databases. Why are these operations crucial? Write the code to insert a record into a MongoDB...
-
You have your own company, the EIE0120, a LLC. This company produces stamped metal parts. You have been asked by a current customer to provide a quotation for a product that is similar to but...
-
What goes into your User Stories/How long are your User Stories? List at least 3 SDLC methodologies. How long does sprints run for? What are the Agile/SCRUM ceremonies? What happens during the Sprint...

Study smarter with the SolutionInn App


