How could you prepare the following ketones by reaction of an acid chloride with a lithium diorganocopper
Question:
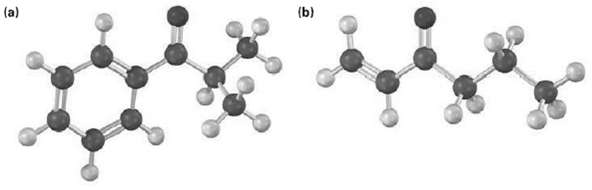
How could you prepare the following ketones by reaction of an acid chloride with a lithium diorganocopper reagent?
Transcribed Image Text:
(b) (a)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 70% (10 reviews)
Two combinations of acid chloride ...View the full answer

Answered By

PALASH JHANWAR
I am a Chartered Accountant with AIR 45 in CA - IPCC. I am a Merit Holder ( B.Com ). The following is my educational details.
PLEASE ACCESS MY RESUME FROM THE FOLLOWING LINK: https://drive.google.com/file/d/1hYR1uch-ff6MRC_cDB07K6VqY9kQ3SFL/view?usp=sharing
3.80+
3+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
How could you prepare the following compounds with benzene as one of the starting materials? (a) (b)
-
How could you prepare the following amides using an acid chloride and an amine or ammonia? (a) CH3CH2CONHCH3 (b) N, N-Diethylbenzamide (c) Propanamide
-
a. Starting with isopropylacetylene, how could you prepare the following alcohols? 1. 2-methyl-2-pentanol 2. 4-methyl-2-pentanol b. In each case a second alcohol would also be obtained. What alcohol...
-
[10 marks] Click the link below to see the code for a program. Your task is as in assignment 3: re-write the code of this program so that, to a user of the program, it works as before, but in your...
-
Even with the popularity of online job-search sites, traditional job-search techniques are still important. What are some traditional sources for finding jobs?
-
a) Could web scraping be a threat to a corporation? Why? b) What are mashups? Give an example. c) What is the difference between a spider and a web scraper? d) Is web scraping ethical, legal,...
-
The annual profits (in thousands of dollars) of a sample of 27 companies listed on a stock exchange Use technology to draw a box-and-whisker plot that represents the data set. 12.86 51.11 13.84 15.96...
-
The unadjusted trial balance of LaBarbara Data at December 31, 2014, appears below. Adjustments had been made until November, but no adjustments for December have been made. Adjustment data: a....
-
The three-part axially loaded member in Fig. P2.2-6 consists of a tubular segment (1) with outer diameter (do)1 1.00 in. and inner diameter (di)1 0.75 in., a solid circular rod segment (2) with...
-
Silver Star Mountain Resort is a ski resort northeast of Vernon in British Columbia. Besides maintaining well-groomed ski runs, the company sells lift tickets, ski lessons, and ski equipment. It...
-
Write the mechanism of the reaction just shown between 3, 4, 5-trimethoxybenzoyl chloride and morpholine to form trimetozine. Use curved arrows to show the electron flow in each step.
-
Write the mechanism of the reaction between p-hydroxyaniline and acetic anhydride to prepare acetaminophen.
-
In Exercises 23 through 30, sketch the indicated level curve f(x, y) = C for each choice of constant C. f(x, y) = xe y ; C = 1, C = e
-
write a by your own words a memo using memo templet to answer the following questions: what is research? what is research question? and please relate the answer of the above questions to...
-
Describe how government intervention affects the supply and demand equilibrium. Refer to the simulation game to explain your responses.
-
Discuss the negative impact of accent stereotypes on immigrants. What steps can you take to advocate against linguistic discrimination ?
-
Explain why collusion is unstable in the one-period Cournot model but may be sustainable in the repeated Cournot model, i.e., in which firms interact over several periods.
-
The nation of Iberia can produce either tablets or cameras. The more it makes of one product, the less it can make of the other. The table shows combinations of the two products it could make from...
-
Are younger New Zealand women becoming sexually active at an earlier age than previous generations of New Zealand women? To shed light on this question, we look at the proportions of New Zealand...
-
What steps must a business take to implement a program of social responsibility?
-
According to MO theory, which molecule or ion has the highest bond order? Highest bond energy? Shortest bond length? 2- 02, 0, 0
-
Indicate whether the following peaks in the mass spectrum of 1-heptanol are odd-electron or even-electron ions. (a) m/z = 83 (b) m/z = 70 (c) m/z = 56 (d) m/z = 41
-
Calculate the energy in kJ mol-1 of the light described in Problem 12.1(b) Blue light with = 4800 A
-
The mass spectrum of methyl isobutyl ether does not show a peak due to inductive cleavage, in contrast to the mass spectrum of di-sec-butyl ether (Eq. 12.31). Use what you know about carbocation...
-
In January 2023, Marty's Fine Pens, a business carried on as a sole proprietorship, sells a limited-edition fountain pen for $125,000. The cost of the pen is $63,000. There is a down payment of...
-
An 90000 loan is amortized by payments of $1850 at the end of every 6 months at a rate of 2% compounded monthly 1. Construct a partial amortization schedule showing the last 2 payments 2. determine...
-
Kelso's has a return on equity of 15.2 percent, a debt-equity ratio of 44 percent, a capital intensity ratio of 1.08, a current ratio of 1.25, and current assets of $138,000. What is the profit...

Study smarter with the SolutionInn App


