How many signals would you expect each of the following molecules to have in its 1H and
Question:
How many signals would you expect each of the following molecules to have in its 1H and 13Cspectra?
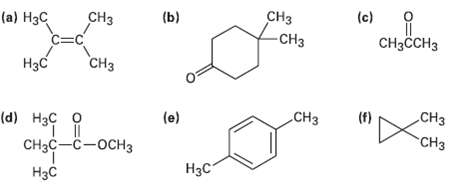
Transcribed Image Text:
(c) CHз -СHз (b) (а) Нзс CHз CHзсCHз CHз Нзс CНз CHз (f) (e) (d) Нзс о "T П CHас —с —оснз "СНз Нзс Нас
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 62% (16 reviews)
a c e HC H3C CC CH3 CH3 13C 2 absorptions 1H 1 absorption CH3CCH3 13C 2 abs...View the full answer

Answered By

Tobias sifuna
I am an individual who possesses a unique set of skills and qualities that make me well-suited for content and academic writing. I have a strong writing ability, allowing me to communicate ideas and arguments in a clear, concise, and effective manner. My writing is backed by extensive research skills, enabling me to gather information from credible sources to support my arguments. I also have critical thinking skills, which allow me to analyze information, draw informed conclusions, and present my arguments in a logical and convincing manner. Additionally, I have an eye for detail and the ability to carefully proofread my work, ensuring that it is free of errors and that all sources are properly cited. Time management skills are another key strength that allow me to meet deadlines and prioritize tasks effectively. Communication skills, including the ability to collaborate with others, including editors, peer reviewers, and subject matter experts, are also important qualities that I have. I am also adaptable, capable of writing on a variety of topics and adjusting my writing style and tone to meet the needs of different audiences and projects. Lastly, I am driven by a passion for writing, which continually drives me to improve my skills and produce high-quality work.
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
How many signals would you expect to find in the 1H NMR spectrum of each of the following compounds?M (a) 1-Bromobutane (c) Butane (b) 1-Butanol (d) 1, 4-Dibromobutane (e) 2, 2-Dibromobutane (g) 1,...
-
How many signals would you expect in the 1 H NMR spectrum of each of the following compounds: (a) (b) (c) (d) (e) (f) CI Br -
-
How many 1H NMR signals would you expect from each of the following compounds? (a) (b) (c) (d) (e) (f) (g) (h) (i) (j) (k) (l) (m) (n) (o) (p) OH CI
-
02. The competitive exclusion principle only applies when the niches of competing species have a high degree of overlap. O True False 54bmit Q3. Four snake species colonize an island at the same...
-
"Integrated firms are more efficient than independent firms if the central office is more efficient than the courts." Explain this statement. To what extent do you agree?
-
Given 2 (1, N = 102) = 3.53, p > .05 where R = 1 and C = 1, (a) Calculate V; (b) Classify the effect as small, medium, or large; and (c) Determine if the researcher should worry about Type II error.
-
As a financial analyst, you want to convince a client of the merits of investing in firms that have just entered a stock exchange, as an initial public offering (IPO). Thus, you gather data on 116...
-
On November 1, 2017, Nordin Inc. (Nordin) issued a $5,000,000 bond with a 6 percent coupon rate and a maturity date of October 31, 2022. Interest is paid semi-annually on April 30 and October 31. The...
-
3. Prepare an unadjusted trial balance as of October 31, 2016. How does grading work? Affordable Realty UNADJUSTED TRIAL BALANCE October 31, 20Y6 ACCOUNT TITLE Score: 40/47 DEBIT CREDIT 1 Cash 2...
-
Barton Company is contemplating the acquisition of the net assets of Crowley Company for $850,000 cash. To complete the transaction, acquisition costs are $15,000. The balance sheet of Crowley...
-
When measured on a spectrometer operating at 200 MHz, chloroform (CHCl3) shows a single sharp absorption at 7.3 . (a) How many parts per million downfield from TMS does chloroform absorb? (b) How...
-
How many absorptions would you expect to observe in the 13C NMR spectra of the following compounds? (a) 1, 1-Dimethylcyclohexane (b) CH3CH2OCH3 (c) tert-Butyl cyclohexane (d) 3-Methyl-i-pentyne (e)...
-
Refer to the data provided in Cornerstone Exercise 10-5. In Cornerstone Exercise 10-5. Bava Inc. produces plastic containers. Each container has a standard labour requirement of 0.02 hours. During...
-
What is the beta of a stock that begins with the same letter as your first name? What is the beta of a stock that begins with the same letter as your last name? Why is it so common to use historical...
-
Why should investors who identify positive-NPV trades be skeptical about their findings if they don't inside information or a competitive advantage? What return should the average investor expect to...
-
Jaycee estimates that the repairs to a Cadillac Escalade damaged in an accident will take 45 hours of labor and $3,500 in parts and materials. The total cost of the repairs is ? The price used to...
-
The Fresh Turkey Company produces two types of turkey cutlets for sale to fast-food restaurants. Each type of cutlet consists of white meat and dark meat. Cutlet 1 sells for $2.97 a pound and must...
-
On May 10, 2016, Oracle stock traded for about $120. All things equal, if Oracle had never had a stock split, what would a share of Oracle have traded for that same day? Round answer to the nearest...
-
Justify whether or not each of the following tables represents the probability distribution of a random variable \(X\) : X P(X) -1 -2 72 47 0.26 0.45 -3 0.22 -4 0.07 X P(X) 0.21 0.20 0.22 0.20 0.23...
-
Use of the contraceptive Depo Provera appears to triple women's risk of infection with chlamydia and gonorrhea , a study reports today. An estimated 20 million to 30 million women worldwide use Depo...
-
An aqueous solution contains 36% HCl by mass. Calculate the molality and mole fraction of the solution.
-
The following proton NMR spectrum is of a compound of molecular formula C3H8O. (a) Propose a structure for this compound. (b) Assign peaks to show which protons give rise to which signals in the...
-
Using a 60-MHz spectrometer, a chemist observes the following absorption: doublet, J = 7 Hz, at 4.00 (a) What would the chemical shift ()be in the 300-MHz spectrum? (b) What would the splitting...
-
A compound (C10H12O2) whose spectrum is shown here was isolated from a reaction mixture containing 2-phenylethanol and acetic acid. (a) Propose a structure for this compound. (b) Assign peaks to show...
-
Activities list and find out the total project duration in total days (You don't need to submit the project diagram, just submit the total days of project duration) What is the Activity Predecessor...
-
Business Value Statement Based on your team review of the client stakeholder environment develop a set of business value propositions. This proposition must indicate what the client offers of value...
-
Tradeoffs existin a variety of different supply chain aspects including transportation. Discuss at least two different tradeoffs that may have to be made when addressing transportation in a global...

Study smarter with the SolutionInn App


