Question: How many stereo isomers exist for this compound? Assign the relative stabilities of each. Is the methyl group axial or equatorial in the more stable
How many stereo isomers exist for this compound? Assign the relative stabilities of each. Is the methyl group axial or equatorial in the more stable conformer of the least stable stereo isomer?
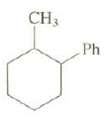
CH3 Ph
Step by Step Solution
3.42 Rating (174 Votes )
There are 3 Steps involved in it
There are four stereoisomers of this compound CH3 CH Ph ... View full answer

Get step-by-step solutions from verified subject matter experts
Document Format (1 attachment)
15-C-O-S (109).docx
120 KBs Word File


