Estimate how much heat is absorbed when 1.00 g of Instant Car Kooler vaporizes. Comment on the
Question:
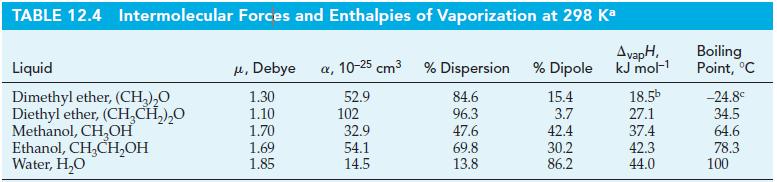
Estimate how much heat is absorbed when 1.00 g of Instant Car Kooler vaporizes. Comment on the effectiveness of this spray in cooling the interior of a car. Assume the spray is 10% C2H5OH(aq) by mass, the temperature is 55 °C, the heat capacity of air is 29 J mol-1 K-1, and use ΔvapH data from Table 12.4.
Table 12.4
Transcribed Image Text:
TABLE 12.4 Intermolecular Forces and Enthalpies of Vaporization at 298 Ka AvapH, kJ mol-¹1 Liquid Dimethyl ether, (CH₂)₂O Diethyl ether, (CH₂CH₂)₂O Methanol, CH₂OH Ethanol, CH₂CH₂OH Water, H₂O μ, Debye a, 10-25 cm³ 1.30 52.9 1.10 1.70 1.69 1.85 102 32.9 54.1 14.5 % Dispersion % Dipole 84.6 15.4 96.3 3.7 42.4 30.2 86.2 47.6 69.8 13.8 18.5b 27.1 37.4 42.3 44.0 Boiling Point, °C -24.8€ 34.5 64.6 78.3 100
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (1 review)
To estimate the amount of heat absorbed when 100 g of Instant Car Kooler vaporizes we need to calcul...View the full answer

Answered By

Wonder Dzidzormenu
As a professional accountant and a teacher, I explain account ing concepts in a more practical way that makes students more connected to the subject.
With over 10 years of teaching accounting , I offer a well constructed , easily understood and in-depth explanations to students questions.
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

General Chemistry Principles And Modern Applications
ISBN: 9780132931281
11th Edition
Authors: Ralph Petrucci, Jeffry Madura, F. Herring, Carey Bissonnette
Question Posted:
Students also viewed these Sciences questions
-
1. a) What is the frequency of a light that has a wavelength of 532 nm? b) Calculate the energy of a photon of light with a wavelength of 532 nm. 2. Consider the transition of the electron in the...
-
Should companies focus on localization? What are the pros and cons of this strategy? Can you think of a company that is focused on localization and achieved success primarily because of this position?
-
A fish at a pressure of 1.1 atm has its swim bladder inflated to an initial volume of 8.16 mL. If the fish starts swimming horizontally, its temperature increases from 20.0C to 22.0C as a result of...
-
Because Natalie has been so successful operating Cookie Creations, Katy would like to have Natalie become her partner. Katy believes that together they will create a thriving cookie-making business....
-
Explain whether the following instrument is negotiable. March 1, 2011 One month from date, I, James Jimson, hereby promise to pay Edmund Edwards: Six thousand, Seven hundred Fifty ($6,750.00)...
-
Recognition of Profit and Balance Sheet Presentation, Percentage-of-Completion On February 1, 2010, Hewitt Construction Company obtained a contract to build an athletic stadium. The stadium was to be...
-
Tyler Companys ASC 932-235 disclosures included the following information: REQUIRED: Using the information for Tyler Company in problems 13, 14, and 15 and in this problem: a. Compute the value of...
-
The city of New Berlin is considering making several of its streets one-way. What is the maximum number of cars per hour that can travel from east to west? The network is shown inFigure. 2 5 0 0 2 2...
-
Problem 4. Consider the following imperfect information game. Notice that both play- ers have two information sets. 5,2 1 a b 2 t 1 d d 4,3 6.0 4,6 0,4 6,0 (a) Find all subgame perfect Nash...
-
A supplier of cylinder gases warns customers to determine how much gas remains in a cylinder by weighing the cylinder and comparing this mass to the original mass of the full cylinder. In particular,...
-
When a wax candle is burned, the fuel consists of gaseous hydrocarbons appearing at the end of the candle wick. Describe the phase changes and processes by which the solid wax is ultimately consumed.
-
XYZ Ltd. a manufacturing company, having an extensive marketing network throughout the country, sells its products throughout four zonal sales offices, viz. A, B, C, and D. The budgeted expenditure...
-
Previously, youth unemployment stood at 22% while the unemployment rate for people in their 30's - 40's averages 4.5% Answer questions a-b to comprehensively analyse the impact of changing...
-
Compare and contrast capitalism with a different economic system and answer the following question accordingly. Describe the organization of the labour process in your chosen system. Describe the...
-
What insights does psychoanalytic social theory offer into the unconscious motivations and desires that underlie individual and collective behaviors, and how can these insights inform our...
-
What is the Marketing budget? Who is the hypothetical target audience? What are the key elements for the visual aspect of the campaign?
-
Kendrick discusses outsourcing and the procurement process. Identify and describe the four components of the procurement process as specified in PMI's PMBOK. Can any of these four components carry a...
-
On December 31, 2012, Berclair Inc. had 200 million shares of common stock and 3 million shares of 9%, $100 par value cumulative preferred stock issued and outstanding. On March 1, 2013, Berclair...
-
The Dow Jones Industrial Average reached a high of $ 7801.63 on December 29, 1997. Recall from Example 18.4 that it reached a high of $ 1003 on November 14, 1972. The Consumer Price Index for...
-
Calculating Interest Rates and Future Values In 1895, the first U.S. Open Golf Championship was held. The winners prize money was $150. In 2006, the winners check was $1,170,000. What was the...
-
Calculating Present Values the first comic book featuring Superman was sold in 1938. In 2005, the estimated price for this comic book in good condition was about $485,000. This represented a return...
-
Calculating Rates of Return although appealing to more refined tastes, art as a collectible has not always performed so profitably. During 2003, Sothebys sold the Edgar Degas bronze sculpture Petite...
-
Internal rate of return method for a service company Vail Resorts, Inc. (MTN), announced a $452,000 million expansion of lodging properties, ski lifts, and terrain in Park City, Utah. Assume that...
-
The accounts in the ledger of Monroe Entertainment Co. are listed in alphabetical order. All accounts have normal balances. Accounts Payable 1,500 Fees Earned 2,400 Accounts Receivable 1,200...
-
On January 1, 2024, National Insulation Corporation (NIC) leased equipment from United Leasing under a finance lease. Lease payments are made annually. Title does not transfer to the lessee and there...

Study smarter with the SolutionInn App


