Question: In Example 15.6, pure-component, liquid-phase adsorption data are used with the extended-Langmuir isotherm to predict a binary-solute data point. Use the following mixture data to
In Example 15.6, pure-component, liquid-phase adsorption data are used with the extended-Langmuir isotherm to predict a binary-solute data point. Use the following mixture data to obtain the best fit to an extended Langmuir-Freundlich isotherm of the form
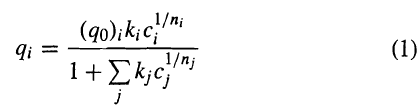
Data for binary-mixture adsorption on activated carbon (1000 m2/g) at 25°C for acetone (1) and propionitrile (2) are asfollows:
1/n (qo);k;c" qi 1+g (1) 1/nj
Step by Step Solution
3.33 Rating (174 Votes )
There are 3 Steps involved in it
From Eq 6 in Example 156 the extended LangmuirFreundlich equations are Fit mixture adsorp... View full answer

Get step-by-step solutions from verified subject matter experts
Document Format (1 attachment)
37-E-C-E-S-P (523).docx
120 KBs Word File


