In light of the fact that tertiary alkyl halides undergo spontaneous dissociation to yield a carbocation plus
Question:
In light of the fact that tertiary alkyl halides undergo spontaneous dissociation to yield a carbocation plus halide ion, propose a mechanism for the followingreaction:
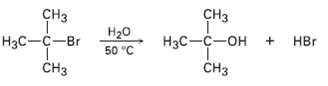
Transcribed Image Text:
CHз сHз Нзс—с—он + НBr CHз Нзс —с—Br Нао 50 "C CHз
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (8 reviews)
CH3 HCCBr CH...View the full answer

Answered By

Branice Buyengo Ajevi
I have been teaching for the last 5 years which has strengthened my interaction with students of different level.
4.30+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
In view of the fact that hot air rises, why does it generally become cooler as you climb a mountain? (Note that air has low thermal conductivity.)
-
In view of the fact that hot air rises, why does it generally become cooler as you climb a mountain? (Note that air has low thermal conductivity.)
-
In Example 26.2, Janets handling of the fact that her data showed 26 volunteers in the control group when there should have been only 25.
-
The balance sheet data for Alans Lightworks, Corp., at August 31, 2012, and September 30, 2012, follow: Requirement 1. The following are three independent assumptions about the business during...
-
Sketch graphs for the relationships described in each of the following problems and select one or more of the families of functions discussed in Section 2.4.4 to represent it. 1. The relationship...
-
Describe the Scenario Manager.
-
Explain the reason the Genetic Information Nondiscrimination Act was enacted.
-
If striving to meet schedule or budget isnt top priority, what is?
-
Describe what the article was about? What stood out the most in the article? How it relates to the fashion industry? Was the article useful in expanding knowledge as it relate to textile? Website...
-
On November 23, 1970, plaintiff Koster and defendant Automark Industries Incorporated (Automark) consummated a five-month course of negotiation by entering into an agreement whereby Automark promised...
-
Tertiary alkyl halides, R 3 CX, undergo spontaneous dissociation to yield a carbocation, R 3 C + , plus halide ion. Which do you think reacts faster, (CH 3 ) 3 CBr or H 2 C = CHC (CH 3 ) 2 Br?...
-
Carboxylic acids (RCO 2 H; p K a 5) are approximately 10 11 times more acidic than alcohols (ROH; pK a 16). In other words, a carboxylate ion (RCO 2 ) is more stable than an alkoxide ion (RO )....
-
Construct comparative boxplots for the thicknesses of new wafers and recycled wafers.
-
4. Tania is a fashion designer her late 30s that plans to retire within 20 years. She enjoys a very "in" lifestyle and likes to hang out with the "in-crowd" in the "in" nightspots whenever she has...
-
What experiences did you have in your early learning years to support diverse cultures in the classroom? Do you feel there was enough exposure to diverse cultures? If not, how can you ensure you...
-
Suppose a(t) = 1.12-0.05. (a) How much interest will be earned during the 5th year on an initial in- vestment of $12? (b) What is the effective annual interest rate during the 5th year?
-
1. Suppose you have access to the following quotes for the : Spot rate: 1.2396 1.2396 180-day forward (outright) rate: 1.2393 What is the corresponding swap rate for the ? In points?
-
Margin requirements in currency futures markets are a form of collateral deposit. True False
-
Joy purchased a home in Maine on December 15, 1999 for $250,000 and lived in it full-time until she purchased a second home in Florida on October 1, 2012 for $380,000. In 2012 and 2013 she continued...
-
Don Griffin worked as an accountant at a local accounting firm for five years after graduating from university. Recently, he opened his own accounting practice, which he operates as a corporation....
-
Draw the Lewis structure for urea, H 2 NCONH 2 , one of the compounds responsible for the smell of urine. (The central carbon atom is bonded to both nitrogen atoms and to the oxygen atom.) Does urea...
-
Which of the following pairs of compounds could be separated by recrystallization or distillation? (a) meso-tartaric acid and (±)-tartaric acid (HOOC-CHOH-CHOH-COOH) (b) (c) (d) CH,CH, O CH,CH...
-
To show that (R)-2-butyl (R, R)-tartrate and (S)-2-butyl (R,R)-tartrate are not enantiomers, draw and name the mirror images of these compounds.
-
The following four structures are naturally occurring optically active compounds. Star the asymmetric carbon atoms in these structures. CHO H CH, COOH OH OH H,N H serine erythrose menthol camphor
-
Textile Crafts Company (TCC) sells craft kits and supplies to retail outlets and through online sites such as Etsy.com. Some of the items are manufactured by TCC, while others are purchased for...
-
Find the missing value in the following table. The following financial statement information is from five separate companies. Company Company Company Company Company A B C D E December 31, 2016...
-
Sunshine Sushi, a Japanese restaurant, has the following adjusted trial balance with accounts listed in alphabetical order. For the bank loan, $60,550 is due in 2024. For Notes receivable, $41,500...

Study smarter with the SolutionInn App


