In a series of observations on the displacement of rubber latex spheres of radius 0.212 urn, the
Question:
In a series of observations on the displacement of rubber latex spheres of radius 0.212 urn, the mean square displacements after selected time intervals were on average as follows:
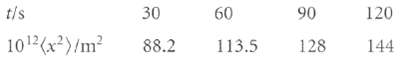
These results were originally used to find the value of Avogadro's constant, but there are now better ways of determining NA' so the data can be used to find another quantity. Find the effective viscosity of water at the temperature of this experiment (25°C).
Transcribed Image Text:
t/s 30 10¹²(x²)/m² 88.2 60 113.5 90 128 120 144
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 68% (16 reviews)
x 2Dt 2183 D Hence n kT Da kTt 3x2 kT 6 206 105 Jm1 2167 ...View the full answer

Answered By

Muhammad Umair
I have done job as Embedded System Engineer for just four months but after it i have decided to open my own lab and to work on projects that i can launch my own product in market. I work on different softwares like Proteus, Mikroc to program Embedded Systems. My basic work is on Embedded Systems. I have skills in Autocad, Proteus, C++, C programming and i love to share these skills to other to enhance my knowledge too.
3.50+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Physical Chemistry questions
-
In a series of studies, Strayer et al. (2003) examined the distraction of drivers in a driving simulation because of cell phone conversations. They hypothesized that driving errors would be made...
-
The Hydrogenics case is the first in a series of four cases that illustrate a comprehensive analysis of an international corporation. In this case the balance sheet will be analyzed with the income...
-
In a series of experiments on the decomposition of dinitrogen pentoxide, N2O5, rate constants were determined at two different temperatures. At 35oC, the rate constant was 1.4 104/s; at 45oC, the...
-
An employee of Gus's Restaurant sustains a work-related injury. The owner of the restaurant is liable for the injury, however, the employee is not covered under the state's workers' compensation...
-
The following table gives weight gain-time data for the oxidation of some metal at an elevated temperature. W (mg/cm2)Time (min) 1.54.................................10...
-
The County Historical Society and Museum is considering holding a picnic as a fundraiser. They plan to charge $15 per ticket. They can rent a facility at a state park for $300. The food would be...
-
Consider the air pollution and mortality data given in Problem 3.15 and Table B. 15 . Problem 3.15 McDonald and Ayers [1978] present data from an early study that examined the possible link between...
-
The adjusted trial balance for Online Advertising Services is presented below. Requirements 1. Prepare the income statement for the year ending December 31, 2016. 2. Prepare the statement of retained...
-
4.4. Solve the problem Ju + uu = 0 (u(x, 0) = g(x) where g(x) = 1 0 TER, t> 0 TER x <0 0 1.
-
The condensed product-line income statement for Blonde Espresso Company is as follows: Fixed costs are 40% of the cost of goods sold and 18% of the selling and administrative expenses. Blonde...
-
Nuclear magnetic resonance can be used to determine the mobility of molecules in liquids. A set of measurements on methane in carbon tetrachloride showed that its diffusion coefficient is 2.05 x...
-
A. Fenghour, W.A. Wake ham, V. Vesovic, I.T.R. Watson, I. Millet, and E. Vogel. Phys. Chem. Ref Data 24, 1649 (1995)) have compiled an extensive table of viscosity coefficients for ammonia in the...
-
The iron-binding site of transferrin in Figure 17-7 can accommodate certain other metal ions besides Fe 3+ and certain other anions besides CO 3 2- . Data are given in the table for the titration of...
-
The open S link shown in the figure has a rectangular cross-section (2 cm x 3 cm). If the applied load is F = 1000 N, determine if point A or point B is more critical for the design (neglect shear...
-
Evaluate the following integrals: 1. Using three and four-point Gauss - Legendre formulas 2. Noting that the exact value is 0.966105, calculate the error. I = 2 1.5 -x dx
-
Q3) (25 points) Surface temperature of a substance is T=1500 K, and spectral emmisivity can be approximated as the figure below. Calculate the total emissivity coefficient. Hemispherical spectral...
-
A single - suction, single - stage, centrifugal pump delivers 2 4 0 lps of sea water ( SG = 1 . 0 1 ) from a source where the water level varies 2 meters from high tide to low tide level.The pump...
-
Consider an investment that covers a 2 - year period. At time t = 0 , an investor bought one share at $ 9 5 . At time t = 1 she purchased another share of the same company at $ 1 2 0 . At the end of...
-
What is the change in value for this security over the four quarters recorded in the highlowclose graph?
-
Research an article from an online source, such as The Economist, Wall Street Journal, Journal of Economic Perspectives, American Journal of Agricultural Economics, or another academic journal. The...
-
Is there a difference between project finance for public works projects like the Big Dig and purely private project finance? If so, what guidelines should be used to help distinguish the two?
-
Which of the following mixtures would you expect to show a positive deviation, a negative deviation, or no deviation (that is, form an ideal solution) from Raoults law? Explain your conclusion. (a)...
-
Calculate the equilibrium constant at 25C and at 150C for each of the following reactions, using data available in Appendix 2A: (a) NH4Cl(s) (b) H(g) + DO(1) NH3(g) + HCl (g) D(g) + HO(1)
-
Calculate the osmotic pressure at 20C of each of the following solutions, assuming complete dissociation for any ionic solutes: (a) 0.050 m C 12 H 22 O 11 (aq); (b) 0.0010 m NaCl(aq); (c) A saturated...
-
The trial balance for a company listed the following account balances at December 31, Year 1, the end of its fiscal year: cash, $36,000; accounts receivable. $31,000; Inventory, $45,000; equipment...
-
A corporation issues 13 %, 15-year bonds with a par value of $570,000 and semiannual interest payments. On the issue date, the annual market rate for these bonds is 11%, which implies a selling price...
-
A production department reports the following conversion costs. Equivalent units of production for conversion total 436,000 units this period. Calculate the cost per equivalent unit of production for...

Study smarter with the SolutionInn App


