Calculate the equilibrium constant at 25C and at 150C for each of the following reactions, using data
Question:
Calculate the equilibrium constant at 25°C and at 150°C for each of the following reactions, using data available in Appendix 2A:![]()
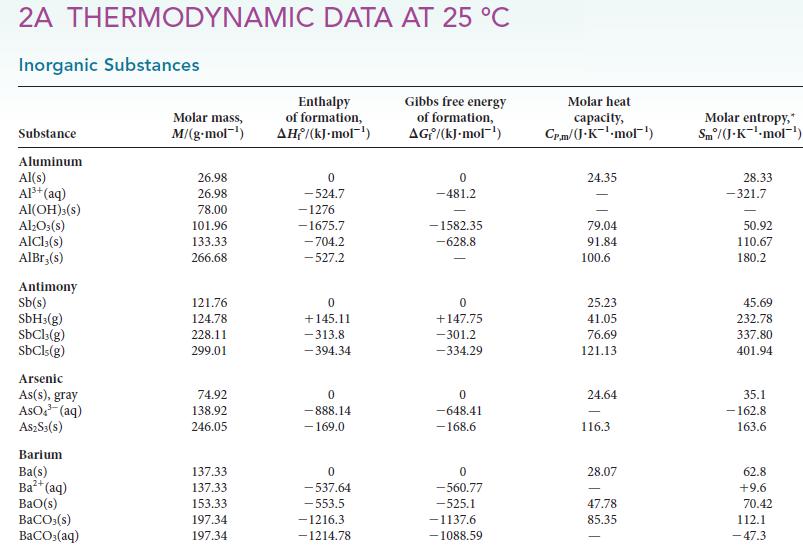
Transcribed Image Text:
(a) NH4Cl(s) (b) H₂(g) + D₂O(1) NH3(g) + HCl (g) D₂(g) + H₂O(1)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (2 reviews)
a At 298 K K 1 X ...View the full answer

Answered By

Utsab mitra
I have the expertise to deliver these subjects to college and higher-level students. The services would involve only solving assignments, homework help, and others.
I have experience in delivering these subjects for the last 6 years on a freelancing basis in different companies around the globe. I am CMA certified and CGMA UK. I have professional experience of 18 years in the industry involved in the manufacturing company and IT implementation experience of over 12 years.
I have delivered this help to students effortlessly, which is essential to give the students a good grade in their studies.
3.50+
2+ Reviews
10+ Question Solved
Related Book For 

Chemical Principles The Quest For Insight
ISBN: 9781464183959
7th Edition
Authors: Peter Atkins, Loretta Jones, Leroy Laverman
Question Posted:
Students also viewed these Sciences questions
-
The budgeted income statement for Barnaby's Hideaway is produced on your Excel spreadsheet. Assume that the following constitute the fixed and vari- able costs for the upcoming year Fixed Costs for...
-
Calculate the equilibrium constant Kc at 25oC from the free-energy change for the following reaction: See Appendix C for data. Zn(s) +2Ag (a)Zn2 (a) Ag(s)
-
Calculate the equilibrium constant at 25C and at 100C for each of the following reactions, using data available in Appendix 2A: (a) 2 CuO (s) 2 Cu(s) + O(g) (b) CH4(g) + H(g) CH6(g)
-
Use the data in the table to complete the following. (a) Make a scatterplot of the data. Estimate a value for b so that f(x) = 0.0002x b models the data. (b) Check the accuracy of f(x). (c) The moon...
-
Repeat Problem 66 for the resistor network shown in Figure. 12 S2
-
Artero Corporation, discussed in Problem 9, is a retailer of toy products. The firms management team recently extended the monthly sales forecasts that were prepared for the last three months of 2011...
-
Show that the variance of the $i^{\text {th }}$ run in a $2^{3}$ design with two replications is \[\frac{\left(y_{i 1}-y_{i 2} ight)^{2}}{2}\] where $y_{i j}$ is $j^{\text {th }}$ observation in...
-
Prescott, Inc. manufactures bookcases and uses an activity-based costing system. Prescotts activity areas and related data follow: Prescott produced two styles of bookcases in October: the standard...
-
Explain how following policy changes will affect the real interest rate and output in the IS-LM framework. Ensure that your answer include an approprtae diagram. i) An increase in the price level ii)...
-
Pizza Corporation acquired 80 percent ownership of Slice Products Company on January 1, 20X1, for $160,000. On that date, the fair value of the noncontrolling interest was $40,000, and Slice reported...
-
Which of the following mixtures would you expect to show a positive deviation, a negative deviation, or no deviation (that is, form an ideal solution) from Raoults law? Explain your conclusion. (a)...
-
Calculate the osmotic pressure at 20C of each of the following solutions, assuming complete dissociation for any ionic solutes: (a) 0.050 m C 12 H 22 O 11 (aq); (b) 0.0010 m NaCl(aq); (c) A saturated...
-
Write each rational expression in lowest terms. x - 81 9-x
-
Give two reasons why Treasury rates are less than other very low risk rates in the United States.
-
What is the formula for the forward price of an investment asset that provides no income?
-
Under what circumstances is the futures price greater than the expected future spot price according to (a) Keynes and Hicks and (b) systematic risk considerations?
-
What is the formula for a forward exchange rate in terms of the spot exchange rate?
-
Explain the two ways derivatives trade in the OTC market?
-
Determine the MARR for a company that can borrow funds at 9% and requires 6% profit margin.
-
If there is an unrealized holding gain on available-for-sale investments, it is reported as?
-
The mechanism of the following transformation involves a carbocation intermediate that rearranges in a way that we have not yet seen. Rather than occurring via a methyl shift or a hydride shift, a...
-
An ideal gas is expanded adiabatically into a vacuum. Decide which of q, w, U and H is positive, negative, or zero.
-
Because U is a state function, (/V (U/T) V ) T = (/T (U/V) T ) V . Using this relationship, show that (CV/V) T = 0 for an ideal gas.
-
difference between index and index fund difference between uits and eitfs give an example from pakistani market
-
provide a 5 line text of the importance of doing a swot analysis when doing a finance analysis for a company
-
The second capital budgeting decision which you were asked to analyze involves choosing between two mutually exclusive projects, S and L, whose cash flows are set forth below: Year 012345 Expected...

Study smarter with the SolutionInn App


