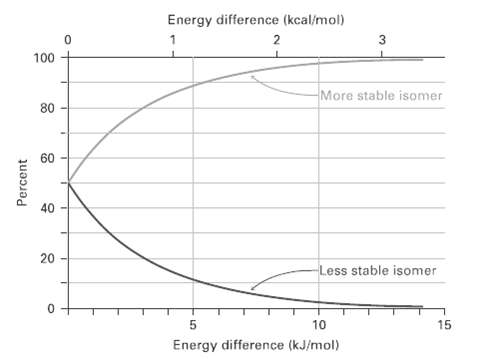
Look at Figure, and estimate the percentages of axial and equatorial conformers present at equilibrium inbromo-cyclohexane. Energy
Question:
Look at Figure, and estimate the percentages of axial and equatorial conformers present at equilibrium inbromo-cyclohexane.
Transcribed Image Text:
Energy difference (kcal/mol) 2 100 More stable isomer 80 60 20 Less stable isomer 10 15 Energy difference (kJ/mol) Percent
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 55% (9 reviews)
Table shows that an axial bromine causes 2 x 10 kJmol of steric strain ...View the full answer

Answered By

JAPHETH KOGEI
Hi there. I'm here to assist you to score the highest marks on your assignments and homework. My areas of specialisation are:
Auditing, Financial Accounting, Macroeconomics, Monetary-economics, Business-administration, Advanced-accounting, Corporate Finance, Professional-accounting-ethics, Corporate governance, Financial-risk-analysis, Financial-budgeting, Corporate-social-responsibility, Statistics, Business management, logic, Critical thinking,
So, I look forward to helping you solve your academic problem.
I enjoy teaching and tutoring university and high school students. During my free time, I also read books on motivation, leadership, comedy, emotional intelligence, critical thinking, nature, human nature, innovation, persuasion, performance, negotiations, goals, power, time management, wealth, debates, sales, and finance. Additionally, I am a panellist on an FM radio program on Sunday mornings where we discuss current affairs.
I travel three times a year either to the USA, Europe and around Africa.
As a university student in the USA, I enjoyed interacting with people from different cultures and ethnic groups. Together with friends, we travelled widely in the USA and in Europe (UK, France, Denmark, Germany, Turkey, etc).
So, I look forward to tutoring you. I believe that it will be exciting to meet them.
3.00+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Look again at textbook Figure 2.2. Trace it onto a piece of scrap paper. Now draw two demand curves on your figure. Draw one that is horizontal at P = $32/MWH and one that is vertical at quantity =...
-
Look at Figure in Chapter 2, which tracks median P/E ratios from 1963 to 2003. Explain why P/E ratios were low in the 1970s and high in the 1960s and 1990s.
-
Look at figure 3.1 on page 60. Precisely which of the six Is were used in case file 3? Which ones were used in case file 1? Were any used in case file 2? INOCULATION One goal of these procedures is...
-
Determine whether the following code fragment takes linear time, quadratic time, or cubic time (as a function of \(n\) ). for (int i = 0; i < n; i++) for (int j = 0; j < n; j++) j) C[i][j] 1.0; if (i...
-
Mark Crane purchased a $1,000 corporate bond five years ago for $1,060. The bond pays 4.5 percent annual interest. Five years later, he sold the bond for $950. Calculate the total return for Mr....
-
How does the rate of population growth influence the level of GDP per person?
-
Consider the automobile gasoline mileage data in Table B.3. a. Build a linear regression model relating gasoline mileage $y$ to vehicle weight $x_{10}$ and the type of transmission $x_{11}$. Does the...
-
Policy Pollsters is a market research firm specializing in political polls. Records indicate in past elections, when a candidate was elected, Policy Pollsters had accurately predicted this 80 percent...
-
Jordan has student loan debt and is worried that it will reduce that amount that can be borrowed for a home. While the percentage of income for a house payment is not a problem, but there is also a...
-
Maggie's Muffins Bakery generated $5,000,000 in sales during 2016, and its year-end total assets were $2,500,000. Also, at year-end 2016, current liabilities were $1,000,000, consisting of $300,000...
-
Why do you suppose an axial cyano (CN) substituent causes practically no 1, 3-diaxial steric strain (0.4 kJ/mol)? Use molecular models to help with your answer.
-
Draw the most stable chair conformation of the following molecules, and estimate the amount of strain in each: (a) trans-1-Chloro-3-methylcyclohexane (b) cis-1-Ethyl-2-rnethylcyclohexane (c)...
-
Explain why a common stock should be evaluated in a portfolio context as opposed to being evaluated in isolation Discuss.
-
How is the recognition of a recovery different from the recognition of a gain? Can a recovery be recorded as income? Why or why not?
-
7. Let L : R R be defined by L(x, y, z) = (x-y, x+2y, z), then: (i) Show that L is a LT. (ii) Find a basis for kernel(L). (iii) Find a basis for range(L). (iv) Is L one-one? (v) Is L onto? (vi) Is L...
-
Your best friend, Margo, knowing that you are taking an income tax class, has asked you for some tax advice. Margo is single and currently has a traditional IRA that she has been making deductible...
-
Use the image to answer the following questions in order: 1. Which of the primate skulls represents a strepsirrhine? 2. Name one characteristic that led you to that conclusion. 3. Which of the...
-
What assumptions and values undergird the Machiavellian leadership style? If power and politics are more important than achievements/performance to impact organizations, does this justify this style...
-
For the data sets below, calculate the following. The linear regression surface. Use matrix regression. The square of the standard error, s 2 e . (a) D = {(42, 79, 1056), (62, 51, 564), (57, 47,...
-
Suppose the government bond described in problem 1 above is held for five years and then the savings institution acquiring the bond decides to sell it at a price of $940. Can you figure out the...
-
A vapor volume of 1.17 L forms when a sample of liquid acetonitrile, CH 3 CN, absorbs 1.00 kJ of heat at its normal boiling point (81.6 C and 1 atm). What is vap H in kilojoules per mole of CH 3 CN?
-
The following reaction does not produce the product shown. (a) Predict the major product from the conditions shown above, and write a detailed mechanism for its formation. (b) What reaction...
-
Predict the products from each of the following reactions. (a) (b) (c) (d) (e) (f) (g) (h) (i) (j) Hl (excess) Hi H,so,,H20 O MeONa O MeOH, cat. H2SO (1) EtSNa (2) H2o HCl (1 equiv.) MeONa (1)EtON...
-
Provide the reagents necessary to accomplish the following syntheses. (a) (b) (c) (d) MeO MeO SEt SEt
-
If you could model yourself after one or more of the historical leaders we discussed in this chapter, whom would you model yourself after? Please learn more about the leader you chose. Identify two...
-
You are a market researcher. You've been asked to compare the way 2 news sources present the same issue for possible consideration by your market research department and public relations staff....
-
Explain about the application software available for Unix operating system?

Study smarter with the SolutionInn App


