Names the following substances, including the cis-or trans-prefix (red-brown =Br): (b) (a)
Question:
Names the following substances, including the cis-or trans-prefix (red-brown =Br):
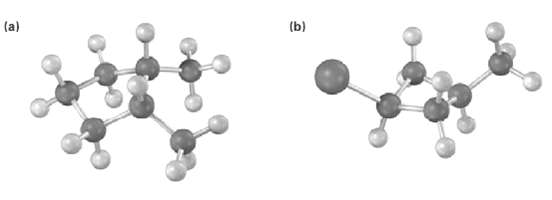
Transcribed Image Text:
(b) (a)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 61% (18 reviews)
CH3 CH3 H cis1...View the full answer

Answered By

Khurram shahzad
I am an experienced tutor and have more than 7 years’ experience in the field of tutoring. My areas of expertise are Technology, statistics tasks I also tutor in Social Sciences, Humanities, Marketing, Project Management, Geology, Earth Sciences, Life Sciences, Computer Sciences, Physics, Psychology, Law Engineering, Media Studies, IR and many others.
I have been writing blogs, Tech news article, and listicles for American and UK based websites.
4.90+
5+ Reviews
17+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Name the following alkenes, including the cis or Tran?s designation: (b)
-
The common names and formulas for several substances are given below. What are the systematic names for these substances? a. Sugar of lead Pb(C2H3O2)2 b. Blue vitriol CuSO4 c. Quicklime CaO d. Epsom...
-
The cis and trans isomers of 2-butene give different Cyclopropane products in the Simmons?Smith reaction. Show the structure of each, and explain the difference. CH2I2, Zn(Cu) cis-CH3CH=CHCH3 CH2I2,...
-
Consider a situation where the marginal damage function is known and equal to D'(E)= d.E. The aggregate marginal abatement cost curve is given by -C'(E)=-bE where is random variable uniformly...
-
Given the information in Question 10, calculate the percent of total return for your $1,350 investment.
-
Consider a linear model to explain monthly beer consumption: beer = (0 + (1inc + (2 price + (3 educ + (4 female + u. E(u|inc, price, educ, female) = 0 Var(u|inc, price, educ, female) = (2 inc2. Write...
-
Describe the measurements that would have to be taken and the equations that would have to be used to determine \(G_{23}, v_{32}\), and \(E_{2}\) for a specially orthotropic, transversely isotropic...
-
Think about the pricing methods described in this chaptermarkup pricing, target-return pricing, perceived value pricing, value pricing, going-rate pricing, and auction-type pricing. As a consumer,...
-
A bond with a maturity of 14 years sells for $1,088. If the coupon rate is 7.2 percent, what is the yield to maturity of the bond?
-
You are interviewing for an entry-level financial analyst position with Wayne Industries. Bruce Wayne, the senior partner, wants to be sure all the people he hires are very familiar with basic...
-
Prostaglandin F2?, a hormone that causes uterine contraction during childbirth, has the following structure. Are the two hydroxyl groups (?OH) on the cyclopentane ring cis or trans to each other?...
-
Each H H eclipsing interaction in ethane costs about 4.0kJ/mol. How many such interactions are present in cyclopropane? What fraction of the overall 115 kJ/mol (27.5kcal/mol) strain energy of...
-
Using logarithmic differentiation, find the derivatives of (a) xe2 In x (b) -esin 2x X
-
How would you describe and occupation? How would you describe a profession? What are the differences?
-
17. How would you describe and occupation? How would you describe a profession? What are the differences?
-
Discuss the due diligence process. Give examples and also send the references used.
-
How do regulators want banks to use derivatives - for what purpose?
-
Explain the five important quantities or measures that are important tomortgage loan analytics.
-
Which of these psychological principles should be the most effective in causing people to change their attitude with regard to smoking? a. Scarcity b. Authority c. Reciprocity d. Consistency e. Liking
-
A firm offers two products for sale. The marginal cost of one product is new zero once the first unit has been produced. The marginal cost of the other product rises as output rises. What would be...
-
One popular demonstration in chemistry labs is performed by boiling a small quantity of water in a metal can (such as a used soda can), picking up the can with tongs and quickly submerging it upside...
-
(a) Outline two methods for preparing isopropyl methyl ether by a Williamson ether synthesis. (b) One method gives a much better yield of the ether than the other. Explain which is the better method...
-
Epoxides can be synthesized by treating halohydrins with aqueous base. Propose a mecha-nism for reactions (a) and (b), and explain why no epoxide formation is observed in (c). (a) (b) (c) CI NaOH H20...
-
Write structures for products A, B, C, and D, showing stereochemistry. (Hint: B and D are stereoisomers.) Ko Br OH (H2) TsCl, pyr OH K CO3
-
A 3.0 resistor is connected across the terminals of a 100 V battery. If 0.50 A of current flows, what is the internal resistance of the battery?
-
The rate of blood flow through the aorta is Q = 100 cm/s. A capillary has an average cross sectional area of Acap = 3 x10 -11 m and supports a blood speed of cap = 1 mm/s. From this information, what...
-
A particle is trapped in a potential well described by U(x)=16-b where U is in joules, x is in meters, and b= 4.0 J. Find the force on the particle when it's at a) x=2.2m and b) x=-1.7m.

Study smarter with the SolutionInn App


