One general reaction of radicals is the coupling of one with another to form a bond, as
Question:
One general reaction of radicals is the coupling of one with another to form a bond, as shown in the following equation: The following coupling reaction gives two products. Show the structures of these products and explain why both are formed.
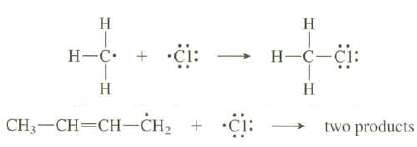
Transcribed Image Text:
H H-C. + -ċi: H CH-CH=CH-CH, + C H H-C-CI: H two products
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 72% (11 reviews)
The odd electron in this radical is in a p orbital so the species ...View the full answer

Answered By

Muhammad Umair
I have done job as Embedded System Engineer for just four months but after it i have decided to open my own lab and to work on projects that i can launch my own product in market. I work on different softwares like Proteus, Mikroc to program Embedded Systems. My basic work is on Embedded Systems. I have skills in Autocad, Proteus, C++, C programming and i love to share these skills to other to enhance my knowledge too.
3.50+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Two flip-flops are connected as shown in the following diagram. The delay represents wiring delay between the two clock inputs, which results in clock skew. This can cause possible loss of...
-
As shown in the following figure, a consumer buys two goods, food and housing, and likes both goods. When she has budget line BL1, her optimal choice is basket A. Given budget line BL2, she chooses...
-
As shown in the following figure, a small metal bar is placed inside container A, and container A then is placed within a much larger container B. As the metal bar cools, the ambient temperature T A...
-
Dominic and Jared were general partnership has equal partners Dominic contributes cash 25,000 securities values at 50,000 and they adjusted basis of 32,000 and equipment valued at 25,000 and an...
-
Waverley Hospital runs an outpatient clinic, Jenny Holt, the Hospital's CEO, is very concerned about cost control and has asked that performance reports be prepared that compare budgeted and actual...
-
Identify the three levels of management in a specific organization by level and title. Be sure to give the organizations name.
-
Water flows in a 10-ft-wide rectangular channel with a flowrate of \(200 \mathrm{ft}^{3} / \mathrm{s}\). Plot the specific energy diagram for this flow. Determine the two possible flowrates when the...
-
Finnish Furniture has experienced a decrease in the demand for tables in Chicago; the demand has fallen to 150 units. What special condition would exist? What is the minimum-cost solution? Will there...
-
The Tom Corp wants to know its cost of capital. Its current capital structure calls for 4 5 % debt, 1 5 % preferred stock and 4 0 % common equity. Initially, common stock will be in the form of...
-
Sentry, Inc. was started on January 1, Year 1. Year 1 Transactions 1. Acquired $20,000 cash by issuing common stock. 2. Earned $62,000 of revenue on account. 3. On October 1, Year 1, borrowed $12,000...
-
Show the three additional resonance structures for anthracene. Discuss whether the experimental bond lengths shown in the following structure are in accord with predictions based on these resonance...
-
Show a MO energy level diagram for the neutral molecule HeH. Use this diagram to explain whether HeH is expected to stable or not?
-
The following transactions were completed by The Corion Gallery during the current fiscal year ended December 31: Feb. 21 Reinstated the account of Tony Marshal, which had been written off in the...
-
You just received an unexpected bonus at work of \($10,000.\) While you plan on using a bit of it to celebrate by purchasing all the accounting books you ever dreamed of, how much should you deposit...
-
Aaron loans Victoria \($10,000\) with interest compounded at a rate of 8% annually. How much will Victoria owe Aaron if she repays the entire loan at the end of five years?
-
A light ray traveling through medium 1 , index of refraction \(n_{1}=1.45\), reaches the interface between medium 1 and medium 2, index of refraction \(n_{2}=1.24\). (a) At what minimum angle with...
-
Data extracted from a year-end balance sheet are shown below. Compute the working capital for this firm. What would the current ratio be, and what is the significance for this firm? Accounts payable...
-
Custom Baseball Hats sells all its merchandise on credit. It has a profit margin of 4 percent, days sales outstanding equal to 60 days (based on a 365-day year), receivables of $\$ 147,945.20$, total...
-
\(\sigma_{1}>\sigma_{2}\) at the \(\alpha=0.05\) level of significance Assume that the populations are normally distributed. Test the given hypothesis.
-
Suppose that you are part of a virtual team and must persuade other team members on an important matter (such as switching suppliers or altering the project deadline). Assuming that you cannot visit...
-
Express 1/(1 + x 2 ) as the sum of a power series and find the interval of convergence.
-
Why is a 1, 3-cis disubstituted cyclohexane more stable than its transisomer?
-
Which is more stable, a 1, 4-trans disubstituted cyclohexane or its cis isomer?
-
Cis-l, 2-Dimnethylcyclohutane is less stable than its transisomer, hut cis-1, 3- dimethylcyclobutanc is more stable than its transisomer. Draw the most stable conformations of both, and explain.
-
Under what conditions would the National Building Code (N.B.C.) be used, as opposed to the Ontario Building Code (O.B.C.)?
-
Each month I have to lie on my expense report in order to get reimbursed for an honest business lunch," is example of?
-
Secondary Analysis: This week visit the Keizer Online Library and find a full-text article on secondary analysis in research. Find a full-text article, download it, highlight it, and upload it to the...

Study smarter with the SolutionInn App


