One of the most historically significant studies of chemical reaction rates was that by M. Bodenstein (z.
Question:
One of the most historically significant studies of chemical reaction rates was that by M. Bodenstein (z. physik. Chem. 29,295 (1899)) of the gas-phase reaction 2 Hl(g) -t H2(g) + I2(g) and its reverse, with rate constants k and k', respectively. The measured rate constants as a function of temperature are
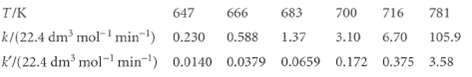
Demonstrate that these data are consistent with the collision theory of bimolecular gas-phase reactions.
Transcribed Image Text:
T/K 647 666 683 700 716 781 k/(22.4 dm³ mol ¹ min¹) 0.230 0.588 1.37 3.10 6.70 105.9 K/(22.4 dm³ mol-¹ min¹) 0.0140 0.0379 0.0659 0.172 0.375 3.58
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 69% (13 reviews)
Linear regression analysis of Inrate constant against 1T yields the following results In k224 dm mol ...View the full answer

Answered By

Asim farooq
I have done MS finance and expertise in the field of Accounting, finance, cost accounting, security analysis and portfolio management and management, MS office is at my fingertips, I want my client to take advantage of my practical knowledge. I have been mentoring my client on a freelancer website from last two years, Currently I am working in Telecom company as a financial analyst and before that working as an accountant with Pepsi for one year. I also join a nonprofit organization as a finance assistant to my job duties are making payment to client after tax calculation, I have started my professional career from teaching I was teaching to a master's level student for two years in the evening.
My Expert Service
Financial accounting, Financial management, Cost accounting, Human resource management, Business communication and report writing. Financial accounting : • Journal entries • Financial statements including balance sheet, Profit & Loss account, Cash flow statement • Adjustment entries • Ratio analysis • Accounting concepts • Single entry accounting • Double entry accounting • Bills of exchange • Bank reconciliation statements Cost accounting : • Budgeting • Job order costing • Process costing • Cost of goods sold Financial management : • Capital budgeting • Net Present Value (NPV) • Internal Rate of Return (IRR) • Payback period • Discounted cash flows • Financial analysis • Capital assets pricing model • Simple interest, Compound interest & annuities
4.40+
65+ Reviews
86+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Physical Chemistry questions
-
One of the most common conflicts in an organization occurs with raw materials and finished goods. Why would finance/accounting, marketing/sales, and manufacturing have disagreements?
-
The prisoners dilemma game is one of the most important models in all of social science: Most games of trust can be thought of as some kind of prisoners dilemma. Heres the classic game: Two men rob a...
-
The guanidino group of arginine is one of the most strongly basic of all organic groups. Explain. NH NI NI
-
Write a method to take an integer array as a parameter and return how many elements having the same digits in each element in the array for example if we have array 1 1 , 4 4 , 1 4 , 2 3 , 1 2 , 5 6...
-
A cylindrical piece of steel 50 mm (2 in.) in diameter is to be quenched in moderately agitated water. Surface and center hard-nesses must be at least 50 and 40 HRC, respectively. Which of the...
-
The section of shaft shown in the figure is to be designed to approximate relative sizes of d = 0.75D and r = D/20 with diameter d conforming to that of standard metric rolling-bearing bore sizes....
-
Analyze the methanol oxidation data in Table B. 20 for multicollinearity. x1 X2 X3 X4 xs 0 454 8.8 3.90 1.30 1.1 474 8.2 3.68 1.16 4.2 524 7.0 2.78 1.25 94.2 503 7.4 2.27 1.57 20.7 493 7.6 2.40 1.55...
-
(a) Plot the above data on a graph. Do you observe any trend, cycles, or random variations? (b) Starting in year 4 and going to year 12, forecast demand using a 3-year moving average. Plot your...
-
How do Lean Management principles integrate with other organizational frameworks, such as Total Quality Management (TQM) and Agile methodologies, to create synergistic approaches for achieving...
-
Shah Ltd (Shah) manufactures a single product, the Choco. The budgeted sales price and production cost per unit of the Choco is as follows: Selling price 360 Variable materials 50 Variable labour 40...
-
For the thermal decomposition Of F2O by the reaction 2 F2O (g) 2 Fl (g) + 0z (g),) Czarnowski and H) Schumacher (Chem. Phys. Lett. 17, 235 (1972)) have suggested the following mechanism: (a) Using...
-
(a) Distinguish between a step and a terrace. (b) Describe how steps and terraces can be formed by dislocations.
-
Describe the company you selected and the products or services it provides.
-
Consider a ball is tossed up vertically. After reaching a max. height of 3 . 0 m , it falls to the floor. The ball comes to rest exactly 1 . 0 m below its starting position. If x is positive upwards,...
-
Europa Ltd just advertized it will pay a 8 , 5 6 euros dividend per share in the end of the year. Current share price is 7 2 , 6 8 euros. An analyst observes that the dividend had been multiplied by...
-
You can afford to pay $400 each month for a car payment. You've found a 4 year loan at 7% interest. How big of a car loan can you afford to take out? Use the TVM Solver on the TI calculator. Round to...
-
Suppose two bonds have the same price and duration. If YTM changes, the price of the higher convexity bond will be _ _ _ _ _ _ _ _ the price of the lower convexity bond.
-
The simplico gold mine has a great deal of remaining gold deposits, and you are part of a team that is considering leasing the mine from its owners for a period of 1 0 years. Gold can be extracted...
-
An entity receives inventories from a counterparty in exchange for a liability based on the price of 4000 of the entitys own shares. At the date of receiving the inventories, the entitys shares have...
-
Based on the scenario described below, generate all possible association rules with values for confidence, support (for dependent), and lift. Submit your solutions in a Word document (name it...
-
Find the area of the shaded region. y y= x+2 y = x= x = 2 1 x+1 X
-
(a) By considering the dependence of the Gibbs free energy of reaction on potential and on temperature, derive an equation for the temperature dependence of E cell . (b) Use your equation to predict...
-
(a) What is the standard cell potential (E cell ) for the reaction below at 298 K? (b) What is the standard cell potential for the reaction at 335 K? (c) What is the cell potential for the reaction...
-
The following items are obtained from a stockroom for the construction of a galvanic cell: two 250-mL beakers and a salt bridge, a voltmeter with attached wires and clips, 200 mL of 0.0080 m CrCl 3...
-
Use the formula for the general term (the nth term) of an arithmetic sequence to find the sixth term of the sequence with the given first term and common difference. a = 10; d=6 86
-
The increasing of the insurance premium has raised a great concern of the public. As the general manager of General Insurance Agency Inc., you would like to use the balanced scorecard to improve the...
-
Your Client Mr. Smith has booked the appointment with your associate and is exploring a variety of Investment and retirement planning options. He wants to better understand the offerings available to...

Study smarter with the SolutionInn App


