Question: One step in the urea cycle for ridding the body of ammonia is the conversion of arginino succinate to the amino acid arginine plus fumarate.
One step in the urea cycle for ridding the body of ammonia is the conversion of arginino succinate to the amino acid arginine plus fumarate. Propose a mechanism for the reaction, and show the structure ofarginine.
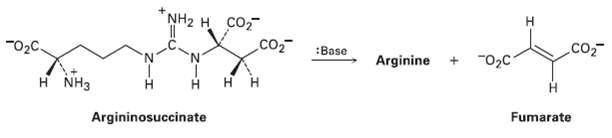
NH2 H CO2 CO "02C. .co :Base Arginine "02C 'N' H NH3 Argininosuccinate Fumarate
Step by Step Solution
★★★★★
3.26 Rating (161 Votes )
There are 3 Steps involved in it
1 Expert Approved Answer
Step: 1 Unlock

0C Base removes a proton yielding an ElcB a... View full answer

Question Has Been Solved by an Expert!
Get step-by-step solutions from verified subject matter experts
Step: 2 Unlock
Step: 3 Unlock
Document Format (1 attachment)
22-C-O-E-R (153).docx
120 KBs Word File


