Order each of the following sets of compounds with respect to SN1reactivity: H3C CH3 C NH2 (a)
Question:
Order each of the following sets of compounds with respect to SN1reactivity:
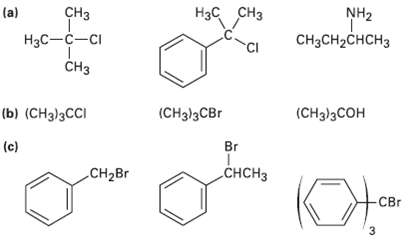
Transcribed Image Text:
H3C CH3 CНз NH2 (a) Нас—с—сi CH3CH2CHCH3 CI CHз (b) (CHз)3СCI (CHд)зСBr (CH)3CОН (c) Br CHCH3 Cн-Br CBr 3
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (16 reviews)
SN1 reactivity Least reactive a b c NH C...View the full answer

Answered By

Asd fgh
sadasmdna,smdna,smdna,msdn,masdn,masnd,masnd,m asd.as,dmas,dma.,sd as.dmas.,dma.,s ma.,sdm.,as mda.,smd.,asmd.,asmd.,asmd.,asm
5.00+
1+ Reviews
15+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Order each of the following sets of compounds with respect to SN2reactivity: CI CH (a) CH3CH2CHCH3 H CH3CH2CH2CI CH (b) CH CH CH CHCCH2Br CHH CH2Br Br CH (c) CH3CH2CH20CH3 CHCH2CH2Br CHCH2CH20Tos
-
Order each of the following sets from the least exothermic electron affinity to the most. a. F, Cl, Br, I b. N, O, F
-
Order the compounds in each of the following sets with respect to increasing acidity: (a) Acetic acid, oxalic acid, formic acid (b) p-Bromobenzoic acid, p-nitro benzoic acid, 2, 4-dinitrobenzoic acid...
-
Abardeen Corporation borrowed $90,000 from the bank on October 1, 2016. The note had an 8percent annual rate of interest and matured on March 31, 2017. Interest and principal were paid in cash on the...
-
How was it possible for a manufacturing plant in North America to have such a turnaround while in the recession?
-
Hierarchical clustering is sometimes used to generate K clusters, K > 1 by taking the clusters at the Kth level of the dendrogram. (Root is at level 1.) By looking at the clusters produced in this...
-
With reference to Exercise 10.50, test the null hypothesis \(p=0.18\) versus the alternative hypothesis \(p eq 0.18\) at the 0.01 level. Data From Exercise 10.50 10.50 In a random sample of 160...
-
A not-for-profit organization receives a restricted gift. When, and in which type of fund, should it recognize the revenue? When, and in which type of fund, should it recognize the related expense?...
-
Which segments of the streaming and television industry (hardware (e.g. Roku), broadcast networks (CBS, NBC), streamers (Netflix, Paramount , Disney ), cable channels (ESPN), content producers...
-
Red's Furniture Manufacturing produces a line of tables and chairs from specialty hardwoods. It makes three different styles of chairs, and each chair takes about the same amount of direct labour...
-
The reactions shown below are unlikely to occur as written. Tell what is wrong with each, and predict the actualproduct. OCCH3)3 . H2CH Br (a) CHCH-CH (CH]l3c (b) Na* - CI LCH3 LCH3 (c) Socil,...
-
Predict the product and give the stereochemistry resulting from reaction of each of the following nucleophiles with (R)-2-bromooctane: (a) CN (b) CH3CO2 (c) CH3S
-
Giving Voice to Values links ethical intent and ethical action to which of the following? a. Kohlbergs Stages of Moral Development b. Rest Four Stage Model c. Jones Moral Intensity Model d. Libby and...
-
From the given dataset and excel output, can you solve the following questions? Salsberry Realty sells homes along the East Coast of the United States. One of the questions most frequently asked by...
-
Erica purchased a franchise agreement to distribute electronic gadgets for 6 years. The agreement cost $2,000,000 and he had to make investments of $825,000 for the first 2 years to set up his...
-
Axel, a young student, has just won $125,000 per year for life in the lottery. He wants to save 80% of this amount for buy a house in 5 years. Subsequently, he plans to save 75% of his lottery...
-
A private equity investor has developed the following financial information for an acquisition target: Expected Projected Projected Projected Projected Projected 2018 2019 2020 2021 2022 2023 Revenue...
-
The strategic alternatives presented by MMI in December 2006 essentially offer two choices. The first option is to adopt greater leverage either directly in BCO or through a leveraged buyout. The...
-
Let F be the accumulated sum, P the principal invested, i the annual compound interest rate, and n the number of years. Which of the following correctly relates these quantities? a. F = P(1+in) b. F...
-
Archangel Corporation prepared the following variance report. Instructions Fill in the appropriate amounts or letters for the question marks in the report. ARCHANGEL CORPORATION Variance...
-
For the reaction 2 A(g) B(g) + 2 C(g), a reaction vessel initially contains only A at a pressure of P A = 255 mmHg. At equilibrium, P A = 55 mmHg. Calculate the value of K p .
-
Predict the major products formed when benzoyl chloride (PhCOCl) reacts with the following reagents. (a) Ethanol (b) Sodium acetate (c) Aniline (d) Anisole and aluminum chloride (e) Excess...
-
Predict the products of the following reactions. (a) Phenol + acetic anhydride (b) Phenol + acetic formic anhydride (c) Aniline + phthalic anhydride (d) Anisole + succinic anhydride and aluminum...
-
Acid-catalyzed transesterification and Fischer esterification take place by nearly identical mechanisms. Transesterification can also take place by a base-catalyzed mechanism, but all attempts at...
-
Microsoft Corporation makes Xbox video game consoles. For Microsoft's financial year YYYY, please make use of the following provided information regarding the inventory of those Xbox consoles:...
-
3. You purchased a 3 year coupon bond one year ago. Its par value is $1,000 and coupon rate is 6%, paid annually. At the time you purchased the bond, its yield to maturity was 6.5%. Suppose you sell...
-
Discuss the following topic(s) in the forum and submit proof of your participation in the online discussions: 1) Professional certification and the practising accountants 2) Cost data and managerial...

Study smarter with the SolutionInn App


